9月 。 2025年22日 10:50 リストに戻る
HPV 28 Genotyping Detection Kit-Taizhou Cowingene Biotech Co., Ltd.|High Sensitivity&Comprehensive Genotype Coverage
その Cowingene HPV 28 遺伝子型検出キット(液体) represents a cutting-edge advancement in molecular diagnostics, specifically designed for the identification of 28 high-risk human papillomavirus (HPV) genotypes. Developed by 台州カウィンジーンバイオテクノロジー株式会社, this kit combines precision, efficiency, and reliability to meet the evolving needs of clinical laboratories and healthcare professionals. This article provides an in-depth analysis of the product's features, technical specifications, applications, and the company's commitment to innovation in molecular diagnostics.
製品概要
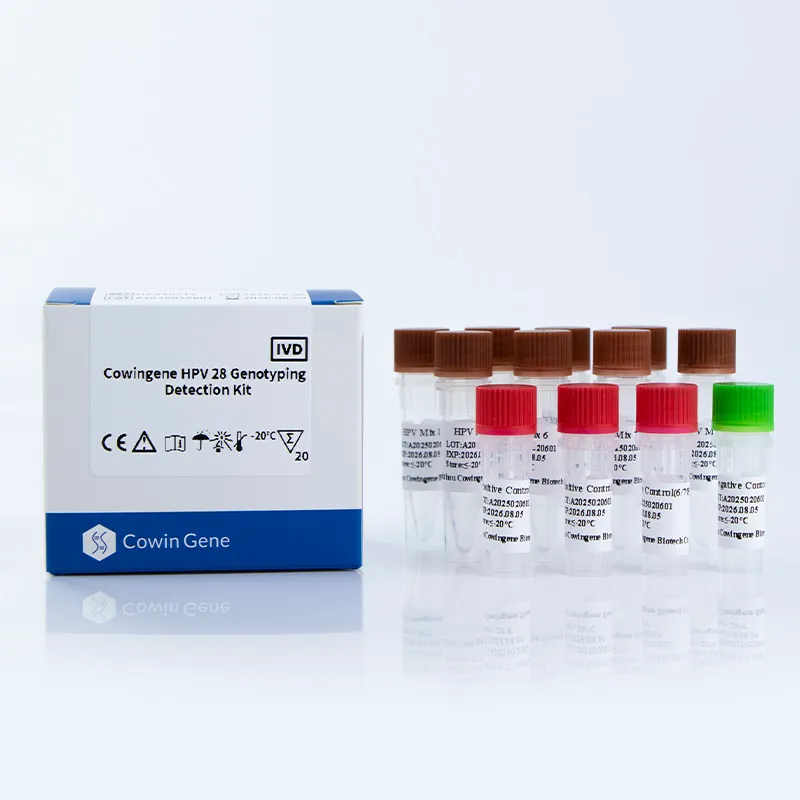
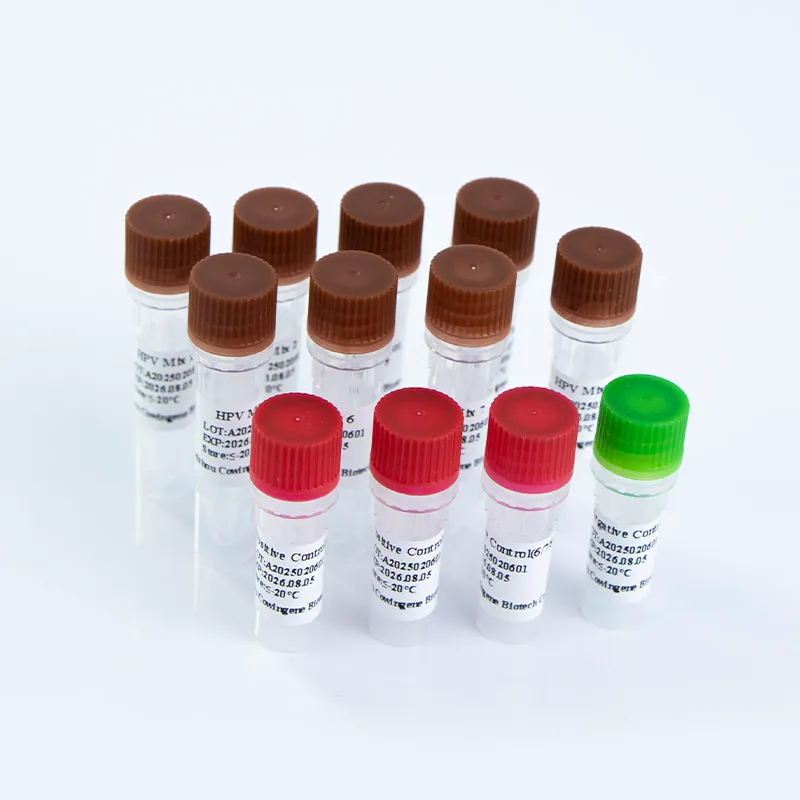
The Cowingene HPV 28 Genotyping Detection Kit is a PCR-based diagnostic tool that enables the simultaneous detection of 28 high-risk HPV genotypes, including 16, 18, 26, 31, 33, 35, 39, 40, 42, 43, 44, 45, 51, 52, 53, 55, 56, 58, 59, 61, 66, 68, 73, 81, 82, and 83. This comprehensive coverage ensures early detection of HPV infections associated with cervical cancer and other related conditions. The kit is validated for use with cervical swab, urine, and self-collected vaginal specimens, offering flexibility in sample collection and processing.
として ヒトパピローマウイルス HPV PCR solution, the kit employs advanced molecular techniques to deliver accurate and reproducible results. Its liquid-based format simplifies workflow, reduces the risk of cross-contamination, and enhances the efficiency of laboratory operations. The product's design aligns with global standards for molecular diagnostics, ensuring compliance with regulatory requirements and clinical best practices.
画像: Read More About hpv detection by pcr
主な特徴と利点
The Cowingene HPV 28 Genotyping Detection Kit stands out in the market due to its exceptional features and advantages, which are critical for modern diagnostic laboratories. Below are some of the key attributes:
- Comprehensive Genotype Coverage: The kit detects 28 high-risk HPV genotypes, including the most prevalent strains linked to cervical cancer, such as 16 and 18. This broad coverage ensures early and accurate identification of at-risk patients.
- 高い感度と特異性: Utilizing optimized primers and probes, the kit delivers high sensitivity (detection of low viral loads) and specificity (minimal cross-reactivity with non-target HPV genotypes), minimizing false positives and negatives.
- Flexible Sample Types: The kit is validated for use with cervical swabs, urine, and self-collected vaginal samples, making it suitable for diverse clinical settings and patient populations.
- Streamlined Workflow: The liquid-based format eliminates the need for complex sample preparation steps, reducing hands-on time and improving laboratory efficiency.
- Compliance with Global Standards: The product adheres to international guidelines for molecular diagnostics, ensuring reliability and acceptance in clinical practice.
画像: Read More About human papilloma virus hpv pcr
Technical Specifications
| パラメータ | 詳細 |
|---|---|
| 製品名 | Cowingene HPV 28 遺伝子型検出キット(液体) |
| メーカー | 台州カウィンジーンバイオテクノロジー株式会社 |
| 検証済み標本 | 子宮頸管スワブ、尿、自己採取膣液 |
| 標的遺伝子型 | 8 tubes covering 28 high-risk HPV genotypes (16, 18, 26, 31, 33, 35, 39, 40, 42, 43, 44, 45, 51, 52, 53, 55, 56, 58, 59, 61, 66, 68, 73, 81, 82, 83) |
| 方法論 | Real-time PCR (qPCR) |
| サンプルの種類 | Liquid-based (no need for DNA extraction in some protocols) |
| 所要時間 | Less than 4 hours (depending on laboratory setup) |
| 保管条件 | 長期保存の場合は-20℃ |
| 貯蔵寿命 | 12 months from the date of manufacture |
画像: Read More About human papilloma virus hpv pcr
Applications and Use Cases
The Cowingene HPV 28 Genotyping Detection Kit is designed for a wide range of applications in clinical and research settings. Its versatility makes it an ideal tool for:
- 子宮頸がんのスクリーニング: The kit is widely used in cervical cancer screening programs to identify high-risk HPV infections, enabling early intervention and reducing mortality rates.
- ポイントオブケア検査: With its streamlined workflow and compatibility with various sample types, the kit is suitable for use in resource-limited settings and mobile clinics.
- 研究と疫学調査: Researchers can utilize the kit to study the prevalence and distribution of HPV genotypes in different populations, contributing to the development of targeted vaccines and therapies.
- Comprehensive Genotyping: The kit's ability to detect 28 high-risk HPV genotypes provides valuable data for patient management and treatment planning.
会社の背景: 台州カウィンジーン生物技術有限公司
2015年に設立され、 台州カウィンジーンバイオテクノロジー株式会社 is a leading biotechnology company specializing in molecular diagnostics and life science research. Based in Taizhou, China, the company has established itself as a trusted provider of innovative diagnostic solutions for infectious diseases, cancer, and genetic disorders.
Cowingene's commitment to quality and innovation is reflected in its state-of-the-art research facilities, rigorous quality control processes, and adherence to international standards. The company's product portfolio includes a wide range of PCR-based assays, immunoassays, and molecular reagents, catering to the needs of clinical laboratories, research institutions, and healthcare providers worldwide.
As a pioneer in the field of molecular diagnostics, Cowingene has received numerous certifications, including ISO 13485:2016 for medical device quality management systems. The company's dedication to continuous improvement and customer satisfaction has earned it a reputation for excellence in the industry.
Scientific Validation and Regulatory Compliance
The Cowingene HPV 28 Genotyping Detection Kit has been rigorously tested and validated to ensure its performance and reliability. According to the 米国国立標準技術研究所 (NIST), molecular diagnostic assays must meet stringent criteria for accuracy, precision, and reproducibility to be considered reliable for clinical use. The Cowingene kit aligns with these standards, as it has undergone extensive validation studies to confirm its analytical performance and clinical utility.
Furthermore, the product complies with the requirements of the U.S. Food and Drug Administration (FDA) そして European Medicines Agency (EMA), ensuring its suitability for use in regulated environments. These certifications underscore Cowingene's commitment to delivering products that meet the highest standards of quality and safety.
結論
その Cowingene HPV 28 遺伝子型検出キット(液体) is a testament to the advancements in molecular diagnostics and the critical role of innovative technologies in improving patient outcomes. With its comprehensive genotype coverage, high sensitivity, and flexibility in sample handling, this kit is an essential tool for cervical cancer screening and HPV research.
As a leader in the biotechnology industry, 台州カウィンジーンバイオテクノロジー株式会社 continues to drive innovation in molecular diagnostics, ensuring that healthcare professionals have access to reliable and efficient solutions. The company's dedication to quality, compliance, and customer satisfaction makes it a trusted partner in the fight against infectious diseases and cancer.
参考文献
1. 米国国立標準技術研究所 (NIST). (n.d.). Molecular Diagnostics Standards. Retrieved from https://www.nist.gov
2. 台州カウィンジーンバイオテクノロジー株式会社. (n.d.). Product Specifications. Retrieved from https://www.cowingene.com
関連している 製品
-
Rapid HPV 16/18/45 Molecular Detection with NATBox for Targeted Cervical Cancer Screening
ニュースApr.24,2026 -
High-Sensitivity HPV (2+16) Molecular Detection for Early Cervical Cancer Screening
ニュースApr.24,2026 -
Comprehensive HPV 28 Genotyping Solution for Advanced Cervical Cancer Screening
ニュースApr.24,2026 -
Comprehensive HPV 15 Genotyping Solution for Accurate Cervical Cancer Risk Assessment
ニュースApr.24,2026 -
Comprehensive 14-Pathogen STI Detection Solution for Accurate Molecular Diagnosis
ニュースApr.24,2026 -
Accurate and Reliable HSV-1/2 Molecular Detection for Clinical Diagnosis
ニュースApr.24,2026








