4-р сар . 24, 2026 13:55 Жагсаалт руу буцах
Comprehensive 14-Pathogen STI Detection Solution for Accurate Molecular Diagnosis
Sexually transmitted infections (STIs) continue to pose a serious global public health challenge due to their high prevalence, diverse pathogen spectrum, and frequently asymptomatic nature. If left undiagnosed or untreated, STIs can lead to severe complications such as infertility, ectopic pregnancy, chronic pelvic inflammation, neonatal infections, and even increased cancer risk. Early, accurate, and comprehensive detection is therefore essential for effective clinical management and disease control.
The Cowingene 14 төрлийн бэлгийн замаар дамжих халдвар илрүүлэх хэрэгсэл (шингэн) is an advanced in vitro diagnostic (IVD) solution designed to provide rapid and reliable qualitative detection of 14 clinically significant STI pathogens. Utilizing multiplex real-time PCR technology, the kit enables simultaneous identification of multiple infectious agents in a single workflow, significantly improving diagnostic efficiency and clinical decision-making.
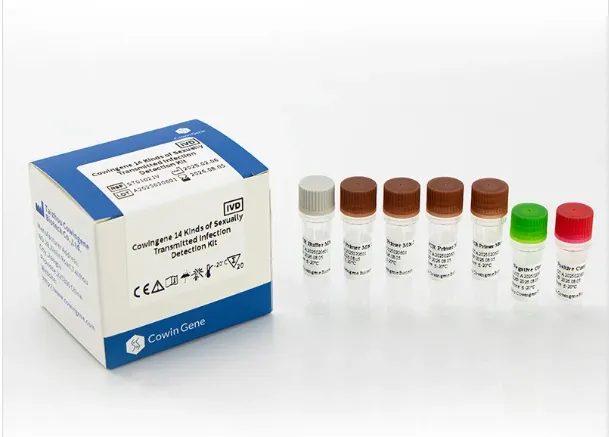
Intended Use: Broad-Spectrum Detection Supporting Clinical Diagnosis
This detection kit is specifically intended for the qualitative detection of the following pathogens associated with genitourinary tract infections:
Chlamydia trachomatis (CT)
Neisseria gonorrhoeae (NG)
Mycoplasma hominis (Mh)
Mycoplasma genitalium (Mg)
Ureaplasma urealyticum (UU)
Ureaplasma parvum (UP)
Herpes simplex virus type 1 (HSV1)
Herpes simplex virus type 2 (HSV2)
Candida albicans (CA)
Gardnerella vaginalis (GV)
Trichomonas vaginalis (TV)
В бүлгийн стрептококк (GBS)
Haemophilus ducreyi (HD)
Treponema pallidum (TP)
The results obtained from this kit serve as important auxiliary evidence for clinicians in the diagnosis and treatment of STIs and related infections. It is suitable for use in hospitals, diagnostic laboratories, and sexual health screening centers, supporting both routine testing and large-scale screening programs.
Flexible Sample Types: Designed for Diverse Clinical Scenarios
To accommodate real-world clinical needs, the kit supports multiple specimen types, including:
Urine samples
Male urethral swabs
Female cervical swabs
Female vaginal swabs
This flexibility allows healthcare providers to select the most appropriate sampling method based on patient condition and testing requirements. The compatibility with self-collected and clinician-collected samples also enhances patient compliance and facilitates broader screening coverage.
Performance Characteristics: High Sensitivity, Specificity, and Stability
High Sensitivity for Early Detection
The kit demonstrates a limit of detection (LoD) of 400 copies/mL across all 14 targets. This high sensitivity ensures reliable detection even in low pathogen load conditions, making it particularly valuable for early-stage infections and asymptomatic carriers.
Excellent Specificity and No Cross-Reactivity
Extensive validation confirms that the kit shows no cross-reactivity with a wide range of non-target microorganisms and nucleic acids, including:
High-risk HPV (16/18)
Escherichia coli
Staphylococcus epidermidis болон Staphylococcus saprophyticus
Lactobacillus species
Adenovirus, Cytomegalovirus (CMV), HIV
Human genomic DNA
This ensures highly accurate pathogen identification and minimizes false-positive results.
Strong Resistance to Interference
The assay maintains stable performance in the presence of common interfering substances such as:
Blood, semen, cervical mucus
White blood cells and mucin
Antibiotics (e.g., levofloxacin, azithromycin, erythromycin, penicillin)
Vaginal care solutions
This robustness reflects real clinical sample conditions and enhances test reliability.
High Precision and Reproducibility
The kit demonstrates excellent repeatability and reproducibility, with coefficient of variation (CV) ≤ 5%, ensuring consistent performance across different runs, operators, and laboratory environments.
Advanced Technical Specifications for Modern Molecular Laboratories
Detection Method: Multiplex real-time PCR (4-tube system)
Detection Channels: FAM, VIC/HEX, ROX, CY5
Compatible Instruments:
ABI 7500
Roche LightCycler 480
Bio-Rad CFX96
Other open PCR platforms
Storage Conditions: ≤ -20°C
Shelf Life: 18 months
Certification: CE-IVD
These specifications ensure seamless integration into existing laboratory workflows while maintaining high analytical performance.
Streamlined Workflow with Supporting Reagents
To optimize the overall testing process, the kit is compatible with a complete range of supporting reagents, including:
DNA/RNA extraction kits
Sample release reagents
Self-collection kits for urine and vaginal samples
This enables a standardized and efficient end-to-end workflow, from sample collection to nucleic acid extraction and amplification, improving laboratory throughput and operational efficiency.
Key Advantages for Clinical and Public Health Applications
Multiplex Detection: Simultaneous identification of 14 pathogens reduces testing time and cost
Comprehensive Coverage: Includes bacteria, viruses, fungi, mycoplasma, and spirochetes
Flexible Sampling: Supports multiple clinically validated specimen types
High Accuracy: Combines high sensitivity and specificity for reliable results
Robust Performance: Resistant to common interfering substances
Wide Compatibility: Works with mainstream real-time PCR instruments
Efficient Workflow: Simplifies laboratory operations and improves turnaround time
Conclusion: Enhancing STI Screening Through Molecular Innovation
The Cowingene 14 Kinds of Sexually Transmitted Infection Detection Kit (Liquid) represents a powerful and reliable molecular diagnostic solution for comprehensive STI screening. By integrating multiplex PCR technology with strong analytical performance and flexible sample compatibility, it addresses the growing demand for accurate, efficient, and scalable diagnostic tools.
This kit not only improves clinical diagnostic accuracy but also supports early intervention strategies, helping to reduce transmission rates and prevent long-term complications. As a result, it plays a critical role in advancing sexual health management and strengthening global public health systems.
Холбоотой БҮТЭЭГДЭХҮҮН
-
Rapid HPV 16/18/45 Molecular Detection with NATBox for Targeted Cervical Cancer Screening
МэдээApr.24,2026 -
High-Sensitivity HPV (2+16) Molecular Detection for Early Cervical Cancer Screening
МэдээApr.24,2026 -
Comprehensive HPV 28 Genotyping Solution for Advanced Cervical Cancer Screening
МэдээApr.24,2026 -
Comprehensive HPV 15 Genotyping Solution for Accurate Cervical Cancer Risk Assessment
МэдээApr.24,2026 -
Accurate and Reliable HSV-1/2 Molecular Detection for Clinical Diagnosis
МэдээApr.24,2026








