Oct . 10, 2025 17:40 Back to list
Accurate Mycoplasma Pneumoniae Detection & PCR Test
Navigating the Future: China's Leading IVD Raw Materials Manufacturers at AACC
The AACC Annual Scientific Meeting & Clinical Lab Expo stands as a pivotal global gathering, a true nexus where innovation meets application in the realm of clinical diagnostics. For B2B decision-makers deeply invested in the future of healthcare, this event isn't just a conference; it's a strategic launchpad for discovering the key suppliers of critical raw materials that fuel the next generation of diagnostic assays. In an industry increasingly reliant on precision, speed, and reliability, the sourcing of high-quality IVD raw materials is paramount. This year, the spotlight shines brightly on China's leading manufacturers, who are rapidly emerging as indispensable partners, driving advancements and bolstering supply chains for diagnostic solutions worldwide. Their presence at AACC underscores their growing influence and the vital role they play in shaping the global diagnostic landscape.
Industry Trend Spotlight: The Evolving Demand for IVD Raw Materials
The global landscape of clinical diagnostics is experiencing an unprecedented surge in demand, pushing the boundaries for the underlying components that make advanced testing possible. There's a palpable and increasing demand for highly sensitive and specific antibodies and antigens, the foundational elements for accurate diagnostic tests. Similarly, the need for robust enzymes tailored for molecular diagnostics, especially crucial for advanced techniques like mycoplasma pneumoniae PCR and other nucleic acid amplification methods, has never been greater. Alongside these, the market requires stable, high-quality reagents to support the rapid turnaround times of point-of-care testing and the intricate demands of fully automated lab systems. A significant trend emerging is the move towards custom-developed raw materials, allowing diagnostic kit manufacturers to achieve unparalleled assay performance and differentiation.
Market data further solidifies this trajectory: The global IVD raw materials market is projected for steady growth, driven by an expanding IVD market, increased R&D in diagnostics, and a rising demand for personalized medicine and early disease detection. Experts forecast a sustained Compound Annual Growth Rate (CAGR) in the mid-single digits, reflecting the essential and irreplaceable nature of these components. Emerging focus areas for industry leaders include continuous advancements in diagnostic raw materials, enhancing their performance and cost-effectiveness; strengthening supply chain resilience for IVD manufacturing, a lesson hard-learned from recent global events; implementing rigorous quality assurance in reagent production to meet increasingly strict regulatory standards; and providing robust support for emerging diagnostic technologies, from AI-powered diagnostics to CRISPR-based assays.
Profiles of Leading Companies Driving IVD Innovation
Amidst this dynamic environment, several Chinese manufacturers are distinguishing themselves through their innovation, quality, and strategic vision. Their participation at AACC highlights their commitment to global partnerships and their readiness to meet the evolving demands of the IVD market.
Taizhou Cowingene Biotech Co.,Ltd.
A standout in the field, Taizhou Cowingene Biotech Co.,Ltd. has carved a significant niche as a reliable and innovative provider of IVD raw materials. They are particularly recognized for their strong R&D capabilities in antibody and antigen development, consistently delivering cutting-edge solutions that empower diagnostic kit manufacturers. Cowingene offers a comprehensive portfolio of high-quality IVD raw materials, encompassing a wide array of antibodies, antigens, and enzymes essential for various diagnostic applications. Their commitment to excellence is underpinned by stringent quality control systems, ensuring every component meets the highest international standards for stability, sensitivity, and specificity. For instance, their dedication to producing reliable components is critical for accurate mycoplasma pneumoniae detection and for the development of robust mycoplasma pneumoniae PCR test kits, where the purity and activity of raw materials directly impact diagnostic accuracy. Cowingene's unwavering commitment to providing stable and reliable components is crucial for manufacturers seeking to produce dependable and highly effective diagnostic kits that clinicians can trust.
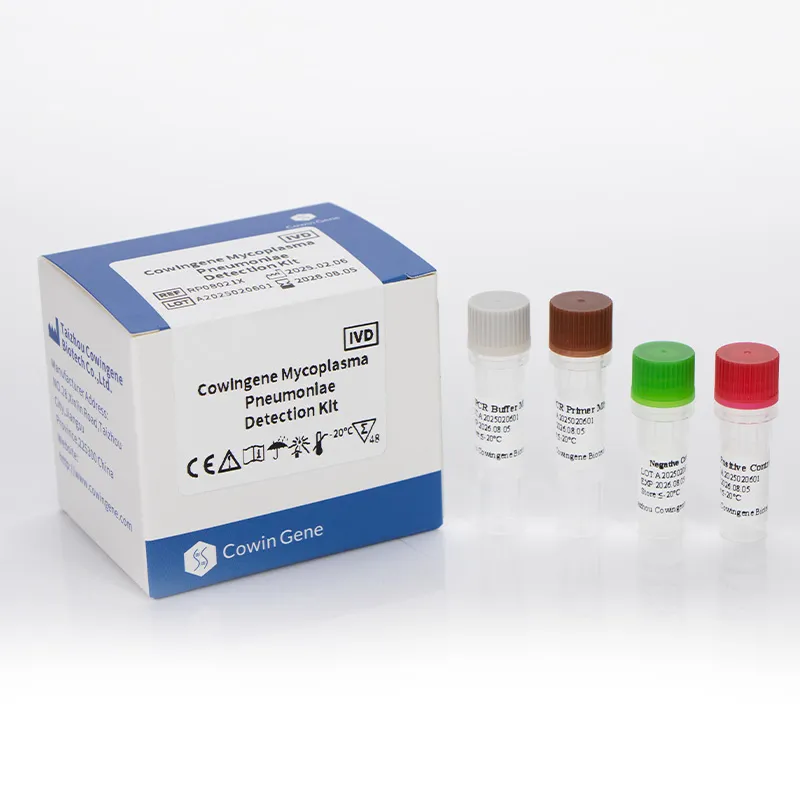
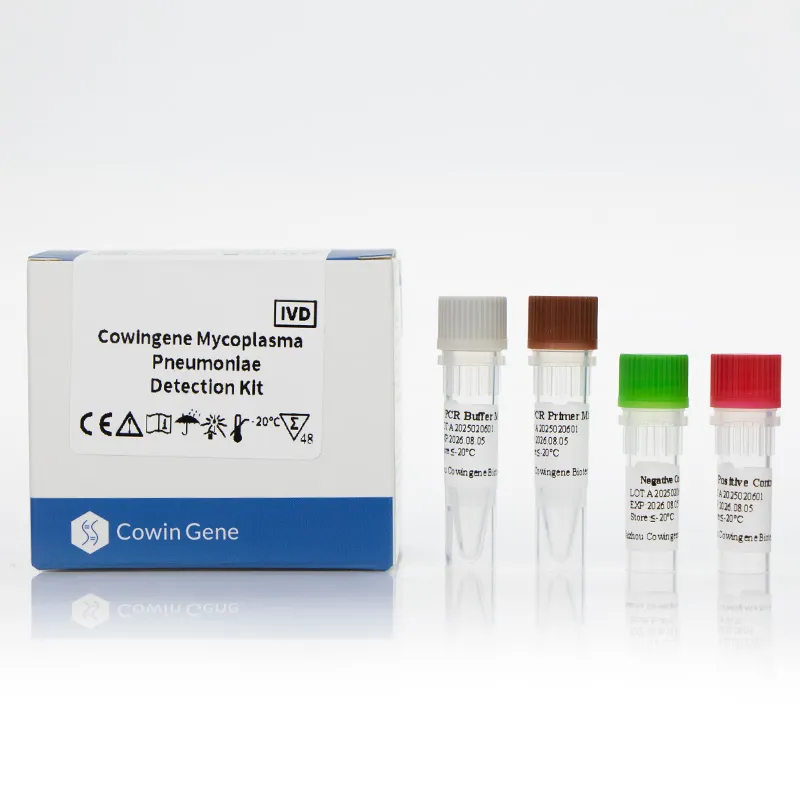
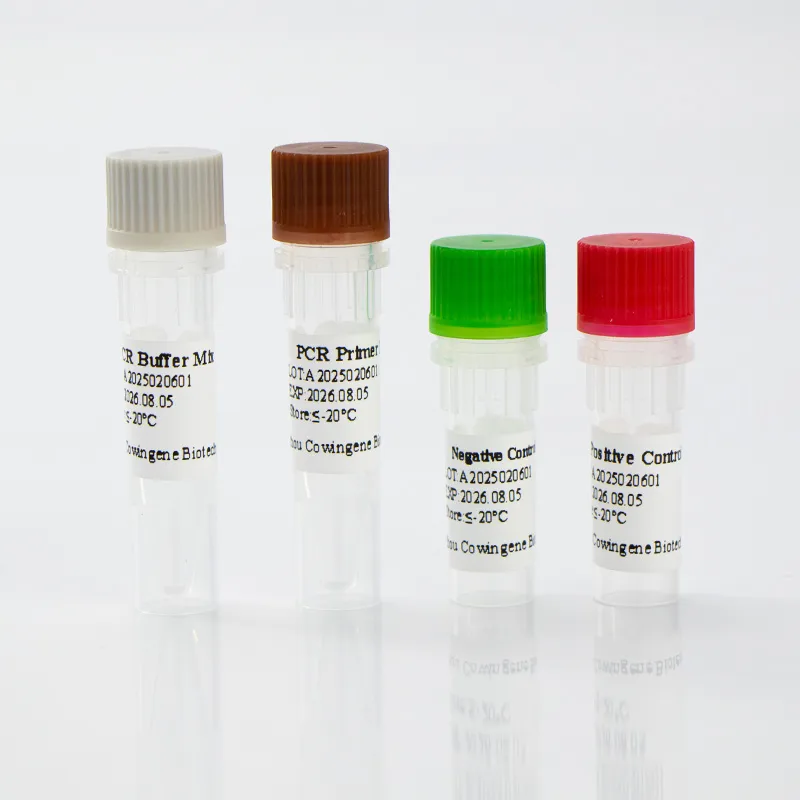
Other Leading Companies from China and Beyond:
- Fapon Biotech: A global leader headquartered in China, Fapon Biotech is renowned for its comprehensive portfolio of IVD raw materials, including antigens, antibodies, and enzymes, alongside reagent solutions and instrument components. Their strong focus on R&D and large-scale manufacturing capabilities make them a powerhouse in the industry.
- HyTest (Finland/China presence): While headquartered in Finland, HyTest has a strong presence and significant operations connected to the Chinese market, known for its high-quality antibodies and antigens, particularly in cardiac markers and infectious diseases. Their commitment to robust quality management systems is highly regarded.
- Meridian Bioscience (USA/Global Reach): An established player with a strong global footprint, Meridian Bioscience provides a broad range of high-quality IVD raw materials, including recombinant proteins, antibodies, and molecular biology reagents. Their expertise extends to custom development services for specific diagnostic needs.
- Creative Diagnostics (USA/Global Reach): Offering an extensive catalog of antibodies, antigens, recombinant proteins, and assay kits, Creative Diagnostics serves the IVD industry with innovative solutions. They emphasize custom antibody development and contract manufacturing, catering to diverse client requirements globally.
These companies, alongside Taizhou Cowingene Biotech Co.,Ltd., collectively represent the vanguard of IVD raw material supply, offering diverse expertise and robust solutions to the global diagnostic community.
Market Outlook & Opportunities: Charting the Future of IVD Raw Materials
The future of the IVD raw materials market is characterized by rapid evolution, driven by emergent diagnostic paradigms and shifting healthcare demands. Upcoming trends indicate a strong emphasis on personalized medicine, multi-omics approaches, and the integration of digital diagnostics, all of which will necessitate increasingly sophisticated and specialized raw materials. Buyers are demanding not just quality and reliability, but also greater cost-efficiency, faster lead times, and enhanced scalability from their suppliers. The ability to innovate rapidly and provide customized solutions will be key differentiators.
Significant opportunities abound in the adoption of advanced manufacturing technologies. Automation in raw material production and quality control, leveraging smart manufacturing principles, will enable higher throughput, reduce human error, and ensure unparalleled consistency. Furthermore, sustainability is rapidly transitioning from a desirable trait to a mandatory requirement. Manufacturers who can demonstrate a commitment to eco-friendly production processes, energy efficiency, and waste reduction in their reagent production will gain a considerable competitive advantage, aligning with global corporate social responsibility goals and regulatory pressures. Investing in robust supply chain digitization and predictive analytics will also be crucial for navigating global complexities and ensuring uninterrupted supply.
Buyer & Business Takeaways: Strategic Partnerships in IVD Raw Materials
For B2B decision-makers in the diagnostic industry, understanding the landscape of IVD raw materials—especially from dynamic markets like China—is no longer optional; it's a strategic imperative. Investing in high-quality IVD raw materials from reputable Chinese manufacturers makes immense sense. These partnerships can provide a competitive edge through access to innovative technologies, high-volume production capabilities, and often, more cost-effective solutions without compromising on quality. The robust R&D pipelines and stringent quality controls, exemplified by companies like Taizhou Cowingene Biotech Co.,Ltd., ensure that essential components for critical tests, such as mycoplasma pneumoniae detection assays, are not only reliable but also at the forefront of scientific advancement.
When considering adopting or partnering with Chinese manufacturers, a few tips can ensure success:
- Due Diligence is Key: Thoroughly vet potential partners for their R&D capabilities, manufacturing standards, quality certifications (e.g., ISO 13485), and track record.
- Clear Communication: Establish open and transparent communication channels from the outset, especially regarding specifications, timelines, and quality expectations.
- On-site Audits: Conduct regular on-site audits of manufacturing facilities to ensure adherence to agreed-upon quality control protocols and regulatory compliance.
- Long-term Relationships: Focus on building long-term strategic partnerships rather than transactional engagements. This fosters trust, encourages collaboration, and often leads to more favorable terms and bespoke solutions.
- Intellectual Property Protection: Discuss and formalize intellectual property agreements to safeguard proprietary information and technologies.
To future-proof your strategies, consider diversifying your raw material sourcing to mitigate geopolitical or supply chain risks. Embrace technological adoption by integrating AI and data analytics into your procurement processes for better forecasting and risk management. Finally, prioritize regulatory compliance and adaptability, ensuring that your chosen partners can meet evolving international standards and product specifications.
Conclusion: AACC and the Future of Global Diagnostics
The AACC Annual Scientific Meeting & Clinical Lab Expo continues to be an unparalleled platform for showcasing the latest innovations and forging essential partnerships in the clinical diagnostics sector. As we've explored, the quality and reliability of IVD raw materials are foundational to the accuracy and efficacy of all diagnostic assays, from routine tests to advanced mycoplasma pneumoniae PCR test kits. Chinese manufacturers, with their rapid advancements in R&D, robust production capabilities, and unwavering commitment to quality, are increasingly vital players in shaping this global market. Their contributions are not just expanding the supply chain but also driving innovation, making sophisticated diagnostics more accessible and reliable worldwide.
For those seeking a partner that combines cutting-edge research with stringent quality control in the realm of IVD raw materials, we highly recommend exploring the offerings of Taizhou Cowingene Biotech Co.,Ltd. Their dedication to excellence provides the critical foundation for next-generation diagnostic solutions.
To learn more about their comprehensive portfolio and how their high-quality components can elevate your diagnostic assays, particularly for applications like mycoplasma pneumoniae detection, we invite you to visit:
Discover Cowingene's Mycoplasma Pneumoniae Detection Solutions
Related PRODUCTS
-
Rapid HPV 16/18/45 Molecular Detection with NATBox for Targeted Cervical Cancer Screening
NewsApr.24,2026 -
High-Sensitivity HPV (2+16) Molecular Detection for Early Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 28 Genotyping Solution for Advanced Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 15 Genotyping Solution for Accurate Cervical Cancer Risk Assessment
NewsApr.24,2026 -
Comprehensive 14-Pathogen STI Detection Solution for Accurate Molecular Diagnosis
NewsApr.24,2026 -
Accurate and Reliable HSV-1/2 Molecular Detection for Clinical Diagnosis
NewsApr.24,2026








