Dec . 02, 2025 16:00 Back to list
Bloodborne Kit – OSHA-Compliant, Complete & Portable
Bloodborne Kit is a key solution in the medical device industry, specifically within in vitro diagnosis and Diagnosis of infectious diseases. This article explores how Taizhou Cowingene Biotech Co.,Ltd. supports professionals with durable, high-performance products, and explains why this product is an ideal choice for businesses in these sectors.
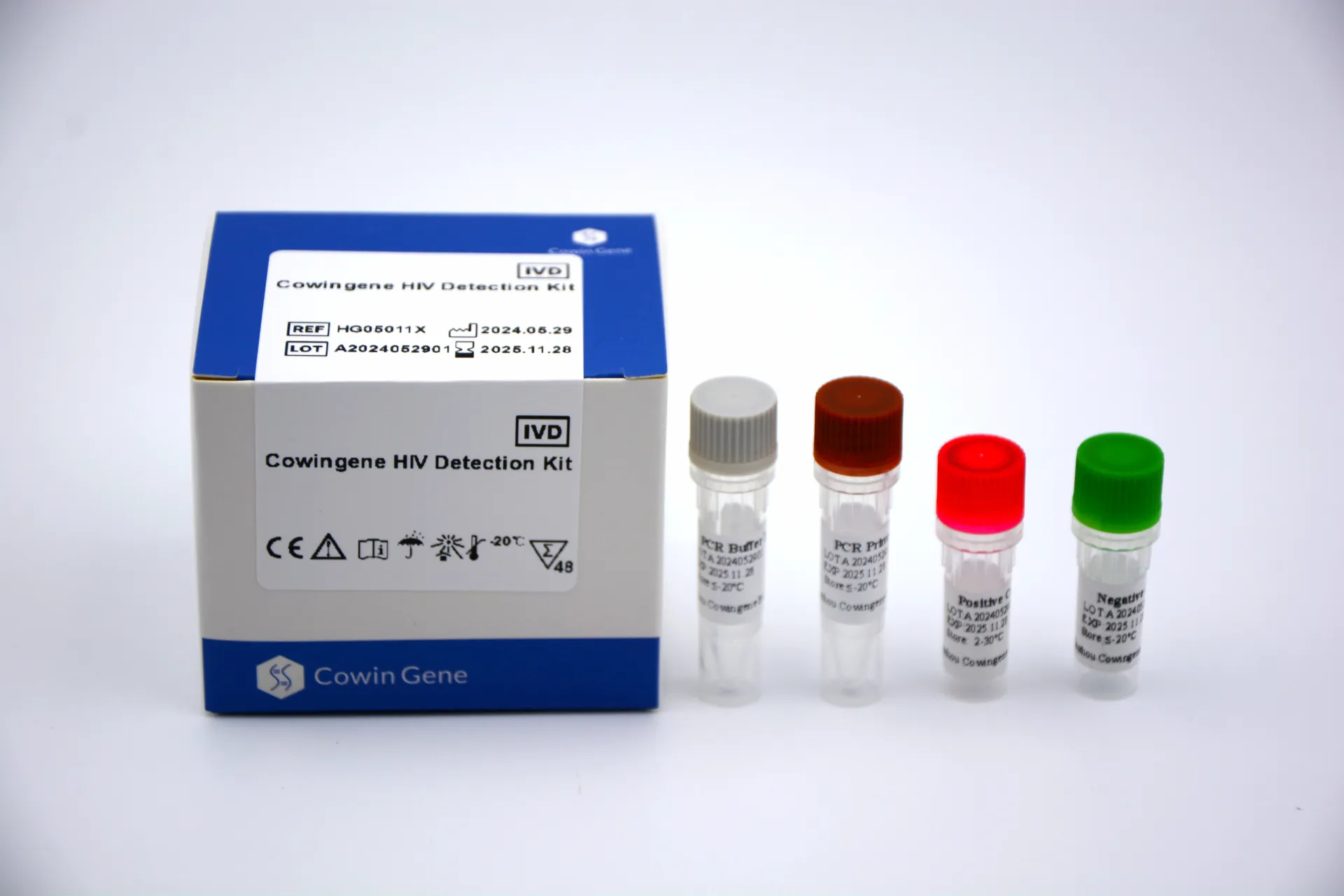

Table of Contents
- Bloodborne Kit Overview
- Benefits & Use Cases of Bloodborne Kit in Diagnosis of infectious diseases
- Cost, Maintenance & User Experience
- Sustainability & Market Trends in medical device
- Conclusion on Bloodborne Kit from Taizhou Cowingene Biotech Co.,Ltd.
Bloodborne Kit Overview
A Bloodborne Kit in the in vitro diagnostics domain is a standardized assay package designed to detect bloodborne pathogens such as HIV, HBV, and HCV from human specimens (whole blood, serum, or plasma). For B2B decision makers in hospitals, reference labs, blood banks, and public health programs, a high-quality bloodborne pathogen kit enables efficient screening, confirmatory testing, and surveillance. Taizhou Cowingene Biotech Co.,Ltd. delivers dependable solutions engineered for consistency, clarity, and operational ease across diverse clinical settings.
- Relevance: Enables rapid and reliable identification of infectious agents in blood, supporting infection control, donor screening, and patient management pathways.
- Technical background: The bloodborne pathogen kit typically includes validated reagents, on-board controls, buffers, sampling accessories, and a clear IFU. Depending on configuration, workflows can support rapid screening or confirmatory algorithms with turnaround ranging from minutes to a few hours.
- Bloodborne pathogen kit contents: test devices/reagents, positive/negative controls, sample diluent/buffer, dropper or transfer pipettes, collection tubes or lancets where applicable, desiccant, and biohazard disposal guidance—designed to streamline end-to-end testing and quality control.
- Manufacturing assurance: Taizhou Cowingene Biotech Co.,Ltd. builds kits under rigorous quality systems and lot-to-lot verification, supporting reproducibility, traceability, and supply continuity for IVD buyers.
Benefits & Use Cases of Bloodborne Kit in Diagnosis of infectious diseases
In real-world practice, a Bloodborne Kit supports multiple care pathways: emergency departments requiring fast HIV/HBV/HCV screens for post-exposure management; antenatal clinics integrating routine testing; blood centers executing donor eligibility assessments; and outreach programs conducting decentralized screening. By standardizing reagents and procedures, organizations improve testing consistency across sites and staff shifts.
- Applications: initial screening, reflex/confirmatory testing workflows, programmatic monitoring, and outbreak response in public health initiatives.
- Competitive advantages: intuitive workflow, clear visual/readable endpoints, robust on-board controls for QC, and comprehensive bloodborne pathogen kit contents that reduce procurement complexity and onboarding time.
- Operational value: consistent performance across diverse matrices (whole blood/serum/plasma, as applicable), streamlined training, and compatibility with standard lab safety practices and documentation.
- Domain expertise: Taizhou Cowingene Biotech Co.,Ltd. focuses on infectious disease IVD solutions, including the HIV detection kit, supporting institutions with technical guidance and reliable supply for program scale-up.
Cost, Maintenance & User Experience
Total cost of ownership for a Bloodborne Kit extends beyond unit price to include training, quality control, wastage, storage, and retesting rates. By providing comprehensive contents and user-friendly IFUs, Taizhou Cowingene Biotech Co.,Ltd. helps reduce invalid results and hands-on time, strengthening ROI. Institutions benefit from simplified inventory (fewer SKUs), predictable performance, and minimized downtime.
- Durability and shelf-life planning: Thoughtful packaging and stability profiles support centralized or distributed testing models, helping reduce spoilage and emergency reorders.
- Maintenance: Most workflows require minimal equipment and maintenance, with routine internal QC via included controls and optional external QC for accreditation needs.
- User experience: Feedback from IVD customers highlights rapid onboarding for new staff, clear result interpretation, and smooth integration into existing SOPs and data capture routines (e.g., barcode-driven sample traceability).
- Strategic procurement: Multi-site healthcare networks can standardize on a single bloodborne pathogen kit, creating economies of scale and improving comparability of results across locations.
Sustainability & Market Trends in medical device
Global demand for infectious disease diagnostics is rising due to expanded screening programs, donor safety mandates, and decentralized care models. Regulatory expectations emphasize quality management, traceability, and consistent performance across diverse settings. Sustainability also matters: health systems increasingly evaluate packaging volumes, cold-chain utilization, and responsible waste handling in their procurement criteria.
- Sustainability focus: Optimized kit formats, right-sized packaging, and clear biohazard disposal guidance help reduce environmental impact without compromising biosafety.
- Digital readiness: Barcoded components and harmonized labeling support LIMS integration and audit trails, aligning with data integrity best practices across laboratories and donor centers.
- Forward-looking partner: Taizhou Cowingene Biotech Co.,Ltd. aligns its bloodborne pathogen kit design with evolving clinical algorithms and procurement standards, helping buyers meet quality, sustainability, and operational targets.
Conclusion on Bloodborne Kit from Taizhou Cowingene Biotech Co.,Ltd.
For organizations seeking dependable screening and surveillance tools, a Bloodborne Kit provides a standardized, efficient approach to detecting HIV and other bloodborne pathogens. Taizhou Cowingene Biotech Co.,Ltd. combines rigorous quality practices with pragmatic design, delivering kits that streamline workflows, support compliance, and scale with program needs. Choose a partner committed to performance, training support, and supply assurance.
- Value recap: Reliable results, simplified procurement, and operational efficiency for medical device / in vitro diagnosis / Diagnosis of infectious diseases.
- Reputation: Taizhou Cowingene Biotech Co.,Ltd. is a trusted manufacturer focused on infectious disease diagnostics and customer-centric service.
- Contact us: email: info@cowingene.com — Visit our website: https://www.cowingene.com
Related PRODUCTS
-
Rapid HPV 16/18/45 Molecular Detection with NATBox for Targeted Cervical Cancer Screening
NewsApr.24,2026 -
High-Sensitivity HPV (2+16) Molecular Detection for Early Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 28 Genotyping Solution for Advanced Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 15 Genotyping Solution for Accurate Cervical Cancer Risk Assessment
NewsApr.24,2026 -
Comprehensive 14-Pathogen STI Detection Solution for Accurate Molecular Diagnosis
NewsApr.24,2026 -
Accurate and Reliable HSV-1/2 Molecular Detection for Clinical Diagnosis
NewsApr.24,2026








