Sep . 08, 2025 12:42 Back to list
Respiratory Panel Test for Rapid, Accurate Pathogen ID
Understanding the Modern Landscape of Respiratory Pathogen Diagnostics
The accurate and rapid identification of respiratory pathogens is paramount in clinical diagnostics and public health. As global health challenges evolve, the demand for sophisticated diagnostic tools, such as the respiratory panel test for comprehensive pathogen detection, continues to surge. These advanced panels are engineered to simultaneously detect multiple viral and bacterial agents responsible for acute respiratory infections, significantly impacting patient management, infection control, and epidemiological surveillance. The shift from single-target assays to multiplexed platforms represents a pivotal advancement, offering unparalleled efficiency and diagnostic breadth.
The emergence of novel pathogens and the persistent circulation of endemic viruses underscore the critical need for diagnostic solutions that are not only sensitive and specific but also rapid and scalable. Traditional methods, while foundational, often lack the turnaround time or multiplexing capabilities required for swift clinical decision-making during outbreaks or severe cases. This has driven the widespread adoption of technologies like the respiratory pathogen panel, which leverages nucleic acid amplification techniques, primarily PCR, to provide definitive results with exceptional speed.
Current Industry Trends Driving Diagnostic Innovation
- Multiplexing Dominance: The trend towards detecting a broader spectrum of pathogens from a single patient sample using a respiratory pathogen panel pcr approach is accelerating. This reduces sample volume requirements, conserves resources, and provides a more complete clinical picture.
- Point-of-Care (POC) Integration: While laboratory-based panels remain the gold standard for comprehensive testing, there is a growing interest in rapid, decentralized diagnostic solutions for initial screening and urgent care settings. Hybrid models combining POC screening with lab-based confirmation are emerging.
- Automation and Throughput: Laboratories are seeking highly automated systems that can handle large volumes of samples with minimal manual intervention, enhancing efficiency and reducing the risk of human error.
- Bioinformatics and Data Integration: Advanced bioinformatics tools are becoming integral for interpreting complex multiplex data, tracking epidemiological trends, and integrating diagnostic results into electronic health records for improved patient management.
- Lyophilized Reagents: The use of lyophilized (freeze-dried) reagents, such as those in the Cowingene Influenza A/B Detection Kit, is a significant trend. This technology enhances kit stability, simplifies storage and transport (often at ambient temperatures), and streamlines laboratory workflows by reducing pipetting steps and potential for contamination, particularly beneficial for rapid deployment and use in diverse settings.
The Advanced Process Flow: Developing a Respiratory Panel Test
The development and manufacturing of a high-quality respiratory panel test for comprehensive pathogen detection involve a rigorous multi-stage process, ensuring accuracy, reliability, and regulatory compliance. This intricate journey transforms scientific understanding into a clinically actionable diagnostic tool. Unlike heavy industrial manufacturing, the "manufacturing process" for diagnostic kits focuses on precision biochemistry, molecular biology, and stringent quality control.
Detailed Manufacturing and Development Stages:
- Target Identification & Primer Design (R&D Phase):
This foundational step involves selecting specific, conserved gene targets for each respiratory pathogen (e.g., Influenza A, B, RSV, SARS-CoV-2, Adenovirus, Parainfluenza, Mycoplasma pneumoniae, Chlamydophila pneumoniae). Highly specific primers and probes are designed to ensure accurate amplification and detection, minimizing cross-reactivity. Bioinformatics tools play a crucial role here, analyzing vast genomic databases.
- Reagent Synthesis & Formulation:
The core components, including high-purity oligonucleotides (primers and probes), DNA polymerase, reverse transcriptase (for RNA viruses), dNTPs, and reaction buffers, are synthesized or acquired. These raw materials undergo stringent quality checks. Formulation involves precise mixing of these reagents to create master mixes that ensure optimal reaction efficiency and sensitivity. For lyophilized kits, like the Cowingene Influenza A/B Detection Kit, this master mix is then prepared for the freeze-drying process.
- Lyophilization (Freeze-Drying) & Kit Assembly:
The formulated master mixes are dispensed into reaction vessels (e.g., PCR tubes, microplates) and subjected to a precisely controlled lyophilization cycle. This process removes water by sublimation, preserving the reagents' activity at ambient temperatures. Lyophilization enhances product stability, extends shelf life, and simplifies cold chain logistics. Post-lyophilization, these individual reaction vessels, along with controls and instructions, are assembled into complete diagnostic kits within a controlled environment.
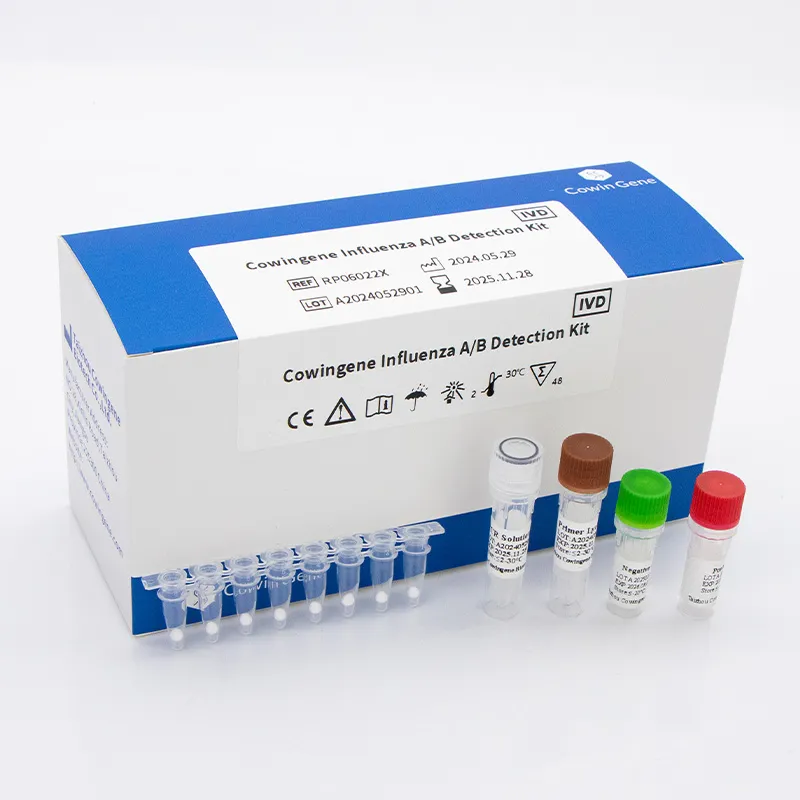
- Quality Control (QC) & Validation:
Each manufactured batch undergoes extensive QC testing. This includes:
- Sensitivity: Determining the lowest detectable concentration of each pathogen.
- Specificity: Ensuring no cross-reactivity with non-target pathogens or human genomic DNA.
- Reproducibility: Verifying consistent results across different runs and operators.
- Stability: Accelerated and real-time stability studies to establish shelf life.
- Limit of Detection (LoD): Quantifying the absolute minimum concentration detectable.
These tests are conducted in accordance with international testing standards such as ISO 13485 for Medical Devices – Quality Management Systems, and often adhere to guidelines set by regulatory bodies like the FDA (for IVD approval) or CE-IVD mark for the European market.
- Packaging & Logistics:
Kits are packaged with appropriate labeling, instructions for use, and necessary accessories. For lyophilized products, ambient shipping is often feasible, reducing logistical costs and environmental impact, while maintaining product integrity. The service life (shelf life) for these kits is typically 12-24 months when stored as recommended.
Target Industries and Advantages:
Respiratory pathogen panel pcr tests are critical for target industries including:
- Clinical Diagnostic Laboratories: Enabling rapid and accurate diagnosis for patient treatment.
- Public Health Agencies: Essential for outbreak surveillance and epidemiological tracking.
- Research Institutions: Facilitating studies on viral and bacterial dynamics.
- Hospitals & Emergency Rooms: Guiding infection control measures and patient isolation protocols.
Key advantages in typical application scenarios include:
- Rapid Diagnosis: Reduces time to result, allowing for prompt therapeutic intervention and preventing unnecessary antibiotic use for viral infections.
- Enhanced Infection Control: Early identification facilitates isolation of infected individuals, minimizing nosocomial transmission.
- Cost-Effectiveness: Multiplexing reduces the need for multiple single-target tests, lowering overall testing costs and improving laboratory efficiency.
- Improved Patient Outcomes: Targeted treatment based on specific pathogen identification leads to better patient recovery and reduced healthcare burden.
Technical Specifications and Performance Parameters
The performance of any respiratory panel test for pathogen detection is defined by its critical technical specifications. These parameters dictate the utility, reliability, and clinical impact of the diagnostic assay. Understanding these specifications is crucial for laboratory professionals and healthcare administrators when evaluating diagnostic solutions.
Key Performance Parameters for Respiratory Pathogen Panels:
| Parameter | Description | Typical Range/Value |
|---|---|---|
| Limit of Detection (LoD) | Lowest concentration of analyte (pathogen) reliably detected. Expressed in copies/mL or TCID50/mL. | 10^1 to 10^3 copies/mL for most targets |
| Sensitivity | Ability of the test to correctly identify positive samples. | Typically >95% (compared to reference method) |
| Specificity | Ability of the test to correctly identify negative samples, avoiding false positives. | Typically >98% (no cross-reactivity with common commensals or non-targets) |
| Turnaround Time (TAT) | Time from sample receipt to result reporting. | 1-4 hours (for multiplex PCR) |
| Sample Types | Validated clinical sample matrices. | Nasopharyngeal swabs, Oropharyngeal swabs, Bronchoalveolar lavage (BAL), Sputum |
| Pathogen Coverage | Number and type of viral/bacterial targets detected. | Typically 15-25 targets (e.g., Flu A/B, RSV, SARS-CoV-2, Metapneumovirus, Adenovirus, Rhino/Enterovirus, Parainfluenza 1-4, M. pneumoniae, C. pneumoniae, B. pertussis) |
| Shelf Life | Duration for which the kit maintains specified performance. | 12-24 months (often at ambient temperatures for lyophilized kits) |
Application Scenarios and Technical Advantages
The versatility of a respiratory panel test for multiple pathogens makes it an indispensable tool across a spectrum of healthcare settings. Its technical advantages address critical gaps in traditional diagnostics, leading to improved clinical and operational efficiencies.
Diverse Application Scenarios:
- Emergency Departments (EDs): Rapid identification of causative agents helps triage patients, determine appropriate isolation protocols, and guide treatment decisions, reducing unnecessary hospitalizations and antibiotic prescriptions.
- Intensive Care Units (ICUs): For critically ill patients with severe respiratory infections, a swift and accurate diagnosis from a respiratory pathogen panel can be life-saving, informing targeted antiviral or antibacterial therapies and preventing secondary infections.
- Pediatric Wards: Young children are particularly vulnerable to severe respiratory infections. Multiplex PCR allows for rapid differential diagnosis, which is crucial as symptoms can be non-specific, preventing complications and aiding in appropriate care.
- Outbreak Management: In institutional settings (e.g., nursing homes, schools) or during community outbreaks, the ability to quickly identify the circulating pathogen(s) using a respiratory pathogen panel pcr test is vital for implementing effective infection control measures and containing spread.
- Immunocompromised Patients: For patients with weakened immune systems, respiratory infections can be particularly severe. A comprehensive panel ensures that even co-infections or less common pathogens are detected, guiding aggressive and appropriate treatment.
Technical Advantages of Advanced Panels:
- Multiplexed Detection: Simultaneously assays for dozens of targets from a single sample, dramatically improving diagnostic yield and reducing the need for sequential testing.
- High Sensitivity and Specificity: Nucleic acid amplification methods (PCR) offer superior analytical performance compared to culture or antigen-based tests, detecting pathogens at very low viral/bacterial loads.
- Rapid Turnaround Time: Results are often available within a few hours, enabling timely clinical interventions that can significantly impact patient outcomes and reduce healthcare costs.
- Differentiation of Co-infections: Capable of identifying multiple pathogens present in a single patient, which is common in respiratory infections and crucial for tailored therapy.
- Reduced Antibiotic Misuse: By clearly identifying viral infections, these panels help reduce inappropriate antibiotic prescriptions, combating antimicrobial resistance.
- Simplified Workflow (with Lyophilized Reagents): Lyophilized kits reduce hands-on time, minimize pipetting errors, and eliminate cold chain storage requirements for reagents, leading to greater laboratory efficiency and accessibility.
Vendor Comparison and Customized Solutions
Selecting the optimal respiratory panel test for a specific laboratory or clinical need requires a thorough evaluation of available vendors. While many offer advanced diagnostic platforms, significant differences exist in their technical specifications, pathogen coverage, throughput, and support infrastructure.
Comparative Analysis of Leading Respiratory Pathogen Panels:
| Feature | Vendor A (e.g., Cowingene - Conceptual Panel) | Vendor B (Competitor) | Vendor C (Competitor) |
|---|---|---|---|
| Pathogen Coverage | 20+ targets (viruses & bacteria, including SARS-CoV-2) | 18 targets (predominantly viruses) | 22 targets (broad viral & bacterial) |
| Assay Technology | Multiplex RT-qPCR (Lyophilized) | Multiplex RT-qPCR (Liquid Reagents) | Microarray-based PCR |
| Turnaround Time | ~2 hours (hands-on time reduced with lyophilized) | ~3-4 hours | ~5-6 hours |
| Sample Input Volume | 200 µL extracted nucleic acid | 200 µL extracted nucleic acid | 500 µL raw sample |
| Regulatory Approvals | CE-IVD, ISO 13485 | CE-IVD, FDA EUA | CE-IVD |
| Storage Conditions | Ambient (2-30°C) | -20°C | 2-8°C |
Note: "Vendor A (Cowingene - Conceptual Panel)" represents the capabilities and advantages typically associated with Cowingene's diagnostic expertise and lyophilized technology, applied to a hypothetical full respiratory panel for illustrative purposes, as the specific Cowingene product linked is for Influenza A/B only. Cowingene's Influenza A/B Detection Kit (Lyophilized) benefits from ambient storage and simplified workflow.
Customized Solutions for Evolving Needs:
Recognizing that no single solution fits all, leading manufacturers often offer customized development and co-development services. This includes:
- Panel Modifications: Adjusting the pathogen target list to specific regional epidemiological needs or research focus. For instance, a panel for a tropical region might include different endemic viruses or bacteria.
- Integration with Existing Platforms: Developing kits compatible with a client's existing PCR instrumentation and laboratory automation systems.
- Format Customization: Offering different reaction formats (e.g., individual tubes, 8-strip tubes, 96-well plates) and reagent presentations (e.g., lyophilized beads, liquid master mixes) to suit diverse laboratory throughput requirements.
- Proprietary Target Inclusion: Incorporating client-specific genetic markers or pathogen variants into the panel design.
- Validation Support: Providing comprehensive validation data and regulatory support for custom panels to ensure compliance and market readiness.
Companies like Cowingene, with their expertise in lyophilized PCR reagent development, are well-positioned to offer such bespoke solutions, providing flexibility while maintaining the high performance and stability benefits of their core technologies.
Application Case Studies and Customer Experience
Real-world applications demonstrate the tangible benefits of implementing an advanced respiratory panel test for comprehensive pathogen identification. These cases highlight improved patient outcomes, operational efficiency, and enhanced public health surveillance.
Case Study 1: Rapid Diagnosis in a Pediatric Hospital
A major children's hospital adopted a comprehensive respiratory pathogen panel for all pediatric patients presenting with acute respiratory symptoms. Prior to implementation, diagnosing the exact cause of respiratory illness often involved multiple sequential tests (e.g., rapid antigen tests for Flu/RSV, followed by individual PCRs), leading to delayed diagnoses, extended hospital stays, and empiric antibiotic use. With the new panel, turnaround time for a broad spectrum of 20+ pathogens was consistently under 3 hours. This enabled clinicians to make precise diagnoses rapidly. For instance, a 2-year-old presenting with bronchiolitis was quickly diagnosed with Human Metapneumovirus (hMPV) and co-infection with Rhinovirus, allowing for immediate discontinuation of broad-spectrum antibiotics and appropriate supportive care. The hospital reported a 25% reduction in average length of stay for pediatric ARI patients and a 30% decrease in unnecessary antibiotic prescriptions within the first year.
Case Study 2: Outbreak Management in an Aged Care Facility
During a severe winter season, an aged care facility experienced a cluster of respiratory illnesses. Initial rapid antigen tests for influenza were negative, leading to uncertainty and challenges in implementing targeted infection control. Upon deploying a respiratory pathogen panel pcr system, within 24 hours, the causative agent was identified as Parainfluenza Virus 3 across multiple residents. This rapid, definitive diagnosis allowed the facility to:
- Implement targeted isolation measures for affected residents.
- Reinforce specific hygiene protocols for Parainfluenza.
- Avoid unnecessary use of antiviral medications meant for influenza.
The swift response, driven by the diagnostic clarity, helped contain the outbreak efficiently, minimizing further morbidity and mortality among the vulnerable resident population.
Customer Feedback Excerpt:
"Integrating Cowingene's lyophilized diagnostic kits, including their influenza A/B product, has streamlined our laboratory workflow significantly. The ambient storage capability alone has revolutionized our inventory management and reduced cold chain costs. More importantly, the reliability and quick turnaround time of their PCR tests have become a cornerstone of our rapid diagnostic capabilities, particularly crucial during flu season. We trust their products for consistent, accurate results."
Commitment to Trust: FAQ, Fulfillment, and Support
Frequently Asked Questions (FAQ):
- Q: What is the typical turnaround time for a respiratory panel test for?
- A: For multiplex PCR panels, results are generally available within 1 to 4 hours from sample reception in the laboratory, depending on extraction method and instrument throughput.
- Q: Can these panels differentiate between viral and bacterial co-infections?
- A: Yes, advanced respiratory pathogen panel tests are designed to detect and differentiate multiple viral and bacterial targets simultaneously, providing a comprehensive picture of the infection.
- Q: Are Cowingene's lyophilized kits stable at ambient temperature?
- A: Yes, Cowingene's lyophilized products, such as the Influenza A/B Detection Kit, are engineered for ambient temperature storage (typically 2-30°C) for their declared shelf life, significantly simplifying logistics and reducing storage costs.
- Q: What regulatory certifications do your products hold?
- A: Our products, including the respiratory pathogen panel pcr components, are manufactured under ISO 13485 certified quality management systems and are CE-IVD marked, ensuring compliance with European directives for in vitro diagnostic medical devices. Specific products may also have local regulatory approvals.
Lead Time and Fulfillment Details:
Cowingene maintains robust manufacturing capabilities and efficient supply chain management to ensure timely delivery of our diagnostic kits.
- Standard Orders: Typically fulfilled within 2-4 weeks, depending on order volume and current stock levels.
- Bulk Orders/Custom Solutions: Lead times will be quoted individually based on project scope, raw material availability, and manufacturing queue. Dedicated project managers ensure clear communication throughout the process.
- Expedited Shipping: Available upon request, subject to additional charges. Our lyophilized products simplify shipping logistics, often allowing for standard (non-refrigerated) transport.
Warranty Commitments:
Cowingene is committed to the quality and performance of its diagnostic products. All kits are warranted to perform according to their published specifications when stored and used as directed within their stated shelf life.
- Product Replacement: In the unlikely event of a manufacturing defect or performance failure, Cowingene will replace the affected product or issue a credit.
- Technical Support: Our dedicated scientific support team is available to assist with troubleshooting, assay optimization, and interpretation of results.
Customer Support Information:
Our expert support team is ready to assist you.
- Email Support: info@cowingene.com
- Phone Support: Available during business hours for technical and order inquiries. Please refer to our website for regional contact numbers.
- Online Resources: Our website features a comprehensive knowledge base, product documentation, and FAQs to provide immediate assistance.
- Dedicated Account Managers: For enterprise clients, a dedicated account manager ensures personalized service and strategic partnership.
References:
- Centers for Disease Control and Prevention (CDC). Respiratory Syncytial Virus (RSV): Clinical Features. Available at: cdc.gov
- World Health Organization (WHO). SARS-CoV-2 and other respiratory pathogens: Laboratory testing, biosafety and biosecurity. Available at: who.int
- Clinical and Laboratory Standards Institute (CLSI). MM03: Molecular Diagnostic Methods for Infectious Diseases. Available at: clsi.org
- International Organization for Standardization (ISO). ISO 13485:2016 Medical devices – Quality management systems – Requirements for regulatory purposes. Available at: iso.org
- European Commission. In vitro diagnostic medical devices. Available at: ec.europa.eu
Related PRODUCTS
-
Cowingene Norovirus Detection Kit
NewsMay.13,2026 -
Cowingene Launches Advanced Lyophilized PCR Kit for Rapid Detection of Group B Streptococcus in Pregnancy Screening
NewsMay.08,2026 -
Cowin Launches Cowingene MG/MH NATBox Detection Kit to Strengthen STI Diagnosis and Point-of-Care Testing Capabilities
NewsMay.08,2026 -
Cowin Introduces Cowingene STI Multiplex Detection Kit (STI 3 NATBox), Advancing Rapid and Accurate Diagnosis of Key Sexually Transmitted Infections
NewsMay.08,2026 -
Comprehensive Overview of the Cowingene Herpes Simplex Virus 1/2 Detection Kit (Liquid)
NewsMay.08,2026 -
Comprehensive Overview of the Cowingene Group B Streptococcus Detection Kit (Lyophilized)
NewsMay.08,2026








