вер . 22, 2025 10:50 Повернутися до списку
HPV 28 Genotyping Detection Kit-Taizhou Cowingene Biotech Co.,Ltd.|HPV PCR Testing&Cervical Cancer Screening
Human papillomavirus (HPV) remains one of the most significant public health challenges globally, with over 100 identified subtypes and 14 classified as high-risk for cervical cancer. The Cowingene HPV 28 Genotyping Detection Kit (Liquid), developed by Taizhou Cowingene Biotech Co., Ltd., represents a breakthrough in molecular diagnostics, offering unparalleled accuracy and efficiency in detecting 28 high-risk HPV subtypes. This article explores the product's advanced features, technical specifications, and real-world applications, while emphasizing its alignment with global health standards.
Overview of the Product
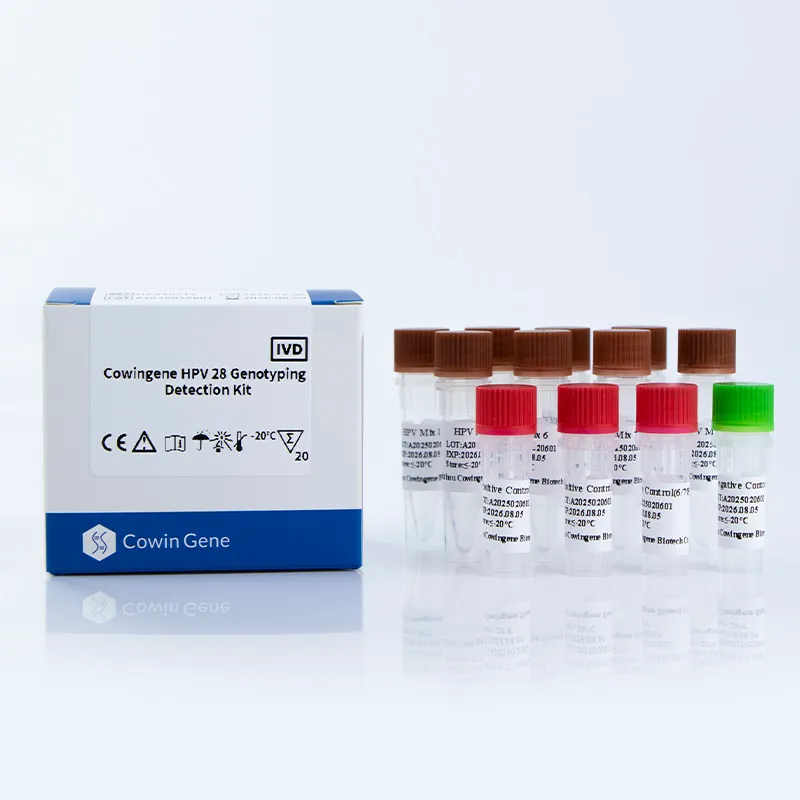
The Cowingene HPV 28 Genotyping Detection Kit is a molecular diagnostic tool designed for the detection and genotyping of human papillomavirus (HPV) in clinical specimens. This kit utilizes polymerase chain reaction (PCR) technology to identify 28 high-risk HPV subtypes, including 16, 18, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 61, 66, 68, 73, 81, 82, 83, and others. The product's versatility is further enhanced by its compatibility with multiple specimen types, making it a valuable asset in cervical cancer screening programs.
Read More About hpv detection by pcr [Сторінка продукту]
Ключові характеристики та переваги
1. Comprehensive Genotyping Coverage
The kit's ability to detect 28 high-risk HPV subtypes sets it apart from conventional assays. This broad coverage ensures that clinicians can identify the most clinically relevant subtypes associated with cervical cancer progression, enabling early intervention and personalized treatment plans.
2. High Sensitivity and Specificity
Utilizing PCR technology, the kit achieves exceptional sensitivity and specificity, minimizing false-negative and false-positive results. This reliability is critical in screening programs where early detection can significantly improve patient outcomes.
3. Versatile Specimen Compatibility
The product is validated for use with cervical swabs, urine, and self-collected vaginal samples, making it accessible for diverse patient populations. This flexibility is particularly important in low-resource settings where traditional sampling methods may be challenging.
Read More About human papilloma virus hpv pcr [Сторінка продукту]
4. Streamlined Workflow and Rapid Results
The kit's design simplifies the diagnostic process, reducing the time required for sample preparation and analysis. This efficiency is crucial in high-volume clinical laboratories, where timely results can impact patient care and resource allocation.
Technical Specifications
| Параметр | Подробиці |
|---|---|
| Перевірені підтипи ВПЛ | 16, 18, 26, 31, 33, 35, 39, 40, 42, 43, 44, 45, 51, 52, 53, 55, 56, 58, 59, 61, 66, 68, 73, 81, 82, 83, 6, 11 |
| Типи зразків | Мазок з шийки матки, сеча, самостійно зібраний вагінальний мазок |
| технології | Генотипування на основі ПЛР |
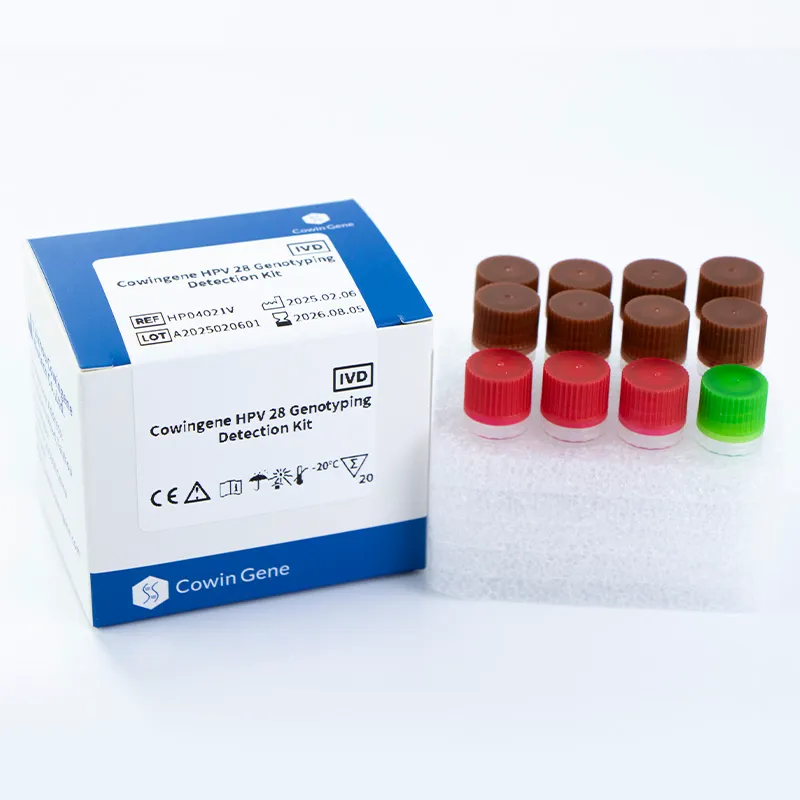
| Компоненти набору | 8 reaction tubes with pre-validated primers and probes |
| Умови зберігання | -20°C протягом 12 місяців |
| Sample Processing Time | Менше 4 годин |
Read More About human papillomavirus pcr [Сторінка продукту]
Applications in Clinical and Public Health Settings
The Cowingene HPV 28 Genotyping Detection Kit is designed for use in a variety of healthcare environments, including:
- Cervical Cancer Screening Programs: The kit's ability to detect high-risk subtypes makes it ideal for large-scale screening initiatives, particularly in regions with limited access to advanced diagnostics.
- Research and Development: Its comprehensive genotyping capability supports epidemiological studies and the development of targeted therapies.
- Primary Care Clinics: The kit's user-friendly design and compatibility with self-collected samples enable early detection in community health settings.
Історія компанії: Taizhou Cowingene Biotech Co., Ltd.
Founded in 2015, Taizhou Cowingene Biotech Co., Ltd. is a leading biotechnology company specializing in molecular diagnostics and infectious disease testing. The company's commitment to innovation is reflected in its state-of-the-art research facilities and partnerships with global health organizations. Cowingene's products are developed in accordance with international standards, ensuring reliability and compliance with regulatory requirements.
Як зазначив в Національний інститут стандартів і технологій (NIST), "Molecular diagnostic tools like the Cowingene HPV 28 Genotyping Kit play a critical role in advancing precision medicine and improving public health outcomes. The integration of PCR-based technologies with rigorous validation protocols ensures that such assays meet the highest standards of accuracy and reproducibility."
Висновок
The Cowingene HPV 28 Genotyping Detection Kit exemplifies the intersection of cutting-edge technology and clinical necessity. By combining advanced PCR-based genotyping with a user-friendly design, the product addresses critical gaps in cervical cancer screening. Its broad coverage, versatility, and reliability make it a valuable tool for healthcare providers, researchers, and public health officials worldwide.
For more information about the product or to explore Cowingene's full range of diagnostic solutions, visit the Cowingene HPV 28 Genotyping Kit page або контакт Taizhou Cowingene Biotech Co., Ltd..
Список літератури
Національний інститут стандартів і технологій (NIST). (nd). HPV Detection and Genotyping Standards. Отримано з https://www.nist.gov/
Пов'язані ПРОДУКЦІЯ
-
Rapid HPV 16/18/45 Molecular Detection with NATBox for Targeted Cervical Cancer Screening
НовиниApr.24,2026 -
High-Sensitivity HPV (2+16) Molecular Detection for Early Cervical Cancer Screening
НовиниApr.24,2026 -
Comprehensive HPV 28 Genotyping Solution for Advanced Cervical Cancer Screening
НовиниApr.24,2026 -
Comprehensive HPV 15 Genotyping Solution for Accurate Cervical Cancer Risk Assessment
НовиниApr.24,2026 -
Comprehensive 14-Pathogen STI Detection Solution for Accurate Molecular Diagnosis
НовиниApr.24,2026 -
Accurate and Reliable HSV-1/2 Molecular Detection for Clinical Diagnosis
НовиниApr.24,2026








