أغسطس . 21, 2025 21:20 Back to list
Fast & Accurate Dengue PCR Test for Early Diagnosis
The Pivotal Role of Dengue PCR Testing in Global Health Surveillance and Diagnostics
Dengue fever, caused by the dengue virus (DENV), presents a significant public health challenge, particularly in tropical and subtropical regions. With an estimated 390 million infections globally each year, accurate and timely diagnosis is paramount for effective patient management, epidemic control, and public health surveillance. Among the array of diagnostic methodologies, the dengue pcr test stands out as a gold standard due to its unparalleled sensitivity, specificity, and ability to detect the virus in the early stages of infection.
The evolution of molecular diagnostics, particularly in the realm of Real-Time Polymerase Chain Reaction (RT-PCR), has revolutionized how we approach vector-borne diseases. For dengue, this means moving beyond serological tests, which often detect antibodies later in the disease course, to directly identifying the viral RNA. This capability is critical for early intervention, differential diagnosis from other febrile illnesses, and for understanding the circulating serotypes during outbreaks.
This comprehensive overview delves into the technical intricacies, operational advantages, and strategic implications of dengue pcr test methodologies. It aims to provide B2B decision-makers, laboratory professionals, and public health experts with insights into current trends, technological parameters, application scenarios, and the critical factors influencing the selection and implementation of these advanced diagnostic solutions.
Technical Foundations: Unpacking Dengue RNA PCR and RT-PCR
The Core Principles of Molecular Detection
A dengue pcr test fundamentally relies on the amplification of specific viral genetic material. Since DENV is an RNA virus, the process typically involves Reverse Transcription Polymerase Chain Reaction (RT-PCR). This molecular technique converts viral RNA into complementary DNA (cDNA) using a reverse transcriptase enzyme, which is then amplified via PCR.
- Reverse Transcription (RT): This initial step is crucial for dengue rna pcr test. Viral RNA extracted from the patient sample (e.g., serum, plasma) serves as a template for synthesizing a cDNA strand. This conversion is necessary because DNA polymerase enzymes used in PCR can only act on DNA templates.
- Polymerase Chain Reaction (PCR): The synthesized cDNA is then subjected to a series of temperature cycles (denaturation, annealing, extension) to exponentially amplify specific target DNA sequences. This amplification generates millions of copies of the target DNA, making even minute amounts of viral RNA detectable.
- Real-Time Detection (qPCR/RT-qPCR): Modern dengue rt pcr test assays incorporate fluorescent dyes or probes that emit signals as DNA amplification occurs in real-time. This allows for quantification of the viral load (the amount of virus in the sample) and eliminates the need for post-PCR gel electrophoresis, significantly reducing turnaround time and contamination risk. The cycle threshold (Ct) value is a key parameter, inversely proportional to the initial amount of target nucleic acid.
Key Technical Parameters and Considerations
- Serotype Detection: Dengue virus has four distinct serotypes (DENV-1, DENV-2, DENV-3, DENV-4). A comprehensive dengue pcr test must be capable of detecting and often differentiating all four serotypes, which is vital for epidemiological surveillance and understanding disease severity, as secondary infections with a different serotype can increase the risk of severe dengue. Multiplex PCR assays are commonly employed for this purpose.
- Sensitivity and Specificity: High sensitivity ensures detection of low viral loads, crucial for early-stage diagnosis. High specificity minimizes false positives by uniquely identifying DENV RNA, distinguishing it from other arboviruses or pathogens. Typical PCR assays boast sensitivities exceeding 95% and specificities near 100%.
- Turnaround Time (TAT): For acute patient management and outbreak response, a rapid TAT is critical. With automated extraction systems and real-time platforms, results for a dengue rt pcr test can often be obtained within 2-4 hours from sample reception.
- Sample Types: The most common sample type is serum or plasma collected during the acute febrile phase (typically within 0-7 days of symptom onset). Other matrices like whole blood, urine, or cerebrospinal fluid (CSF) may also be used in specific clinical contexts.
- Detection Window: PCR assays offer the broadest detection window during the viremic phase, typically days 1-7 post-symptom onset, overlapping with the period of peak viral load. This window is critical for early diagnosis when antibody responses might still be absent or too low for detection by serological tests (e.g., IgM/IgG ELISA).
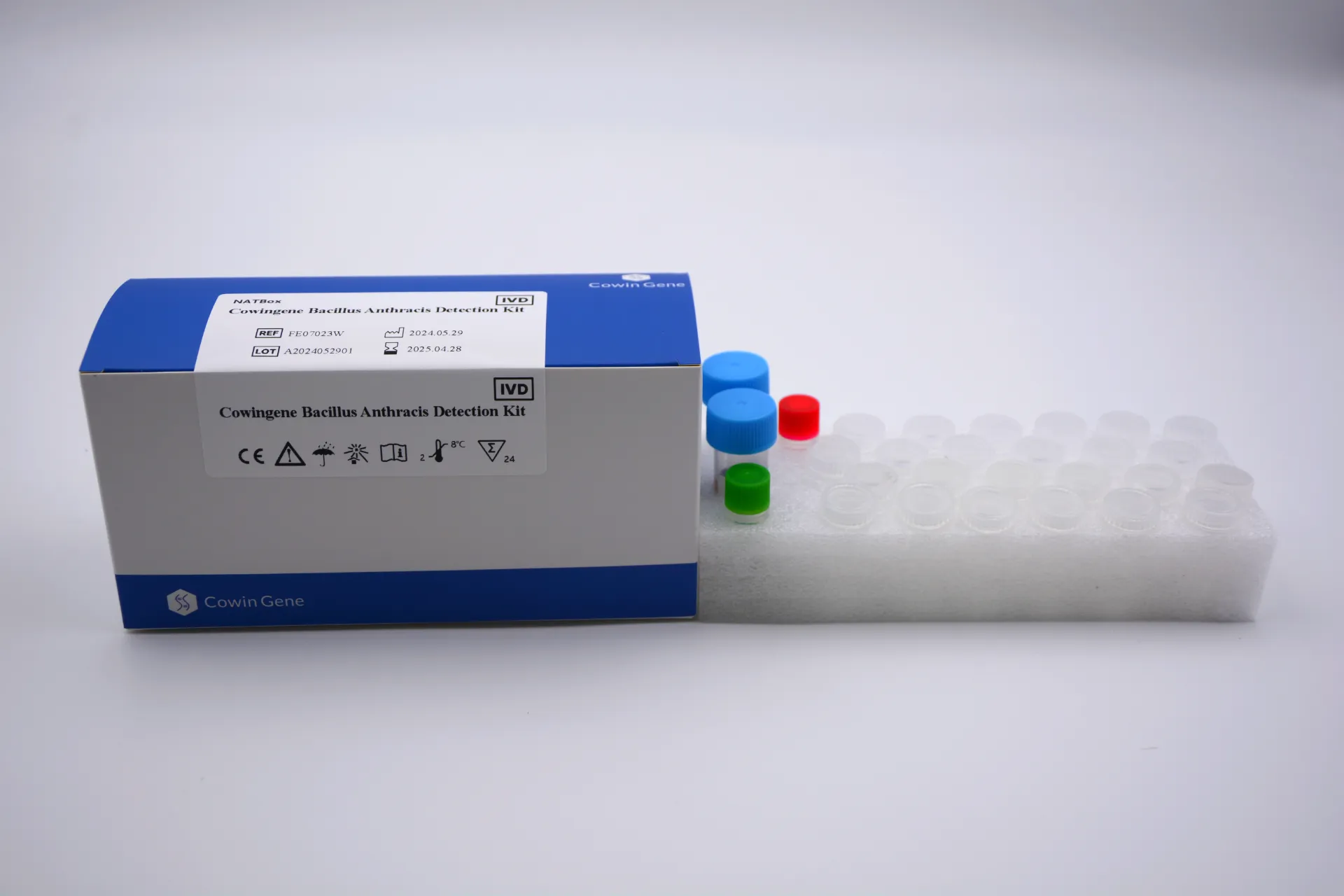
The Dengue PCR Test Workflow: From Sample to Result
Detailed Process Flow and Quality Control
The workflow for a dengue pcr test kit, from sample reception to result interpretation, is a meticulously designed process that prioritizes accuracy, reproducibility, and efficiency. This process is applicable whether utilizing a sophisticated automated system or a more manual laboratory setup.
1. Sample Collection and Reception:
- Materials: Blood collection tubes (serum separator or EDTA plasma), sterile needles.
- Process: Venipuncture by trained healthcare professionals. Samples are transported to the lab under specific temperature conditions (typically 2-8°C) to maintain nucleic acid integrity.
- Standards: Adherence to CLSI (Clinical and Laboratory Standards Institute) guidelines for sample collection and handling. Proper labeling and documentation are crucial for traceability.
2. Nucleic Acid Extraction:
- Materials: Specialized extraction kits (magnetic bead-based, silica-column based), lysis buffer, wash buffer, elution buffer. Consumables are typically high-grade plastics, certified RNase/DNase-free.
- Process: This critical step isolates viral RNA from clinical samples while removing inhibitors that could interfere with PCR. It involves cell lysis, binding of nucleic acids to a solid phase, washing away impurities, and elution of pure RNA. Automated extraction platforms significantly improve throughput and reduce hands-on time and contamination risk.
- Manufacturing Standards for Kits: Reagents used in extraction kits are manufactured under stringent ISO 13485 (Medical devices – Quality management systems) certified conditions, ensuring lot-to-lot consistency and performance. Raw materials are often sourced from certified suppliers.
3. Reverse Transcription and PCR Amplification:
- Materials: RT-PCR master mix containing reverse transcriptase, DNA polymerase (e.g., Taq polymerase), dNTPs, buffer, target-specific primers, and fluorescent probes (for real-time PCR). Reaction vessels are typically 96-well or 384-well PCR plates made from high-grade, optically clear polypropylene.
- Process: The extracted RNA is combined with the master mix in a thermal cycler. The machine performs precise temperature cycles for RT and PCR. The dengue rt pcr test process involves initial reverse transcription, followed by cycles of denaturation (95°C), annealing (50-65°C), and extension (72°C).
- Quality Control during Kit Manufacturing: Primers and probes are synthetically manufactured with extremely high purity standards. Enzymes are produced recombinantly and undergo rigorous quality checks for activity, purity, and absence of contaminating nucleases. Each batch of a dengue rna pcr test kit is tested for sensitivity, specificity, and stability across a range of viral loads and against known dengue serotypes.
4. Data Analysis and Interpretation:
- Equipment: Real-time PCR instrument (thermocycler with fluorescence detection capabilities) and associated software.
- Process: The software analyzes fluorescence signals to generate amplification curves and Ct values. Internal controls are critical for validating the extraction and amplification steps, ensuring no false negatives due to inhibition or RNA degradation. Positive and negative controls are run with every batch.
- Detection Standards: Results are interpreted based on predefined Ct thresholds. A detectable amplification curve below the threshold indicates a positive dengue pcr test result. Automated interpretation streamlines the process, reducing human error.
Product Lifespan and Applicable Industries
The shelf life of dengue pcr test kits typically ranges from 12 to 24 months when stored under recommended conditions (-20°C for reagents, 2-8°C for buffers). The robust manufacturing processes, including lyophilization (freeze-drying) for some reagents, contribute to their extended stability and ease of transport. These kits are primarily utilized in:
- Clinical Diagnostic Laboratories: For rapid and accurate diagnosis of suspected dengue cases, guiding patient management.
- Public Health and Reference Laboratories: For epidemiological surveillance, outbreak investigation, and confirming results from rapid diagnostic tests.
- Research Institutions: For studies on dengue pathogenesis, vaccine development, and antiviral drug screening.
- Blood Banks: For screening blood donations in endemic areas to prevent transfusion-transmitted dengue.

Advantages and Application Scenarios of Dengue PCR Tests
Unrivaled Diagnostic Precision
The distinct advantages of a dengue pcr test over traditional diagnostic methods, such as NS1 antigen detection or IgM/IgG antibody assays, are manifold. Primarily, its ability to directly detect viral genetic material offers a critical edge in the early acute phase of infection (typically 0-7 days post-symptom onset), even before the host immune response generates detectable antibodies.
- Early Detection: This is arguably the most significant advantage. Early and accurate diagnosis facilitates prompt clinical management, reducing the risk of progression to severe dengue and improving patient outcomes. It also allows for timely implementation of public health measures to control disease spread.
- High Specificity: PCR targets unique viral sequences, minimizing cross-reactivity with other flaviviruses (e.g., Zika, Chikungunya, Yellow Fever) that often circulate in the same regions, thus reducing false positives and aiding in precise differential diagnosis.
- High Sensitivity: Dengue RNA PCR test can detect very low viral loads, making it reliable even when viral concentrations are low. This is particularly beneficial for cases presenting with atypical symptoms or in pre-symptomatic stages during surveillance efforts.
- Serotype Identification: Multiplex dengue rt pcr test assays can simultaneously detect and differentiate all four DENV serotypes. This information is crucial for epidemiological monitoring, understanding outbreak dynamics, and predicting potential severity, as sequential infections with different serotypes can increase the risk of severe dengue.
- Viral Load Quantification: Real-time PCR provides quantitative results (Ct values), allowing for estimation of viral load. While not yet a routine clinical parameter for dengue, viral load kinetics can potentially be used in research settings to monitor disease progression or treatment response.
Key Application Scenarios
- Acute Clinical Diagnosis: For patients presenting with acute febrile illness in dengue-endemic areas, a dengue pcr test provides definitive diagnosis in the early viremic phase, guiding immediate clinical decisions and supportive care.
- Outbreak Investigation and Surveillance: Public health agencies leverage dengue pcr test for rapid identification of circulating serotypes, tracking disease spread, and informing vector control strategies during outbreaks.
- Differential Diagnosis: In regions where dengue co-circulates with other arboviruses (e.g., Zika, Chikungunya), PCR assays enable precise differentiation, which is essential for accurate patient management and public health reporting.
- Travel Medicine: For travelers returning from dengue-endemic regions with febrile symptoms, PCR testing offers reliable early diagnosis, distinguishing dengue from other travel-related illnesses.
- Blood Product Screening: In countries with a high burden of dengue, nucleic acid amplification tests (NAT), including dengue pcr test, are increasingly being implemented to screen blood donations, minimizing the risk of transfusion-transmitted infections and enhancing blood safety.
- Research and Vaccine Development: PCR serves as a fundamental tool for virological studies, evaluating vaccine efficacy, and monitoring antiviral drug candidates.
In critical application scenarios, such as rapidly evolving outbreaks, the speed and accuracy of a dengue pcr test are indispensable. For instance, in a surge of unexplained febrile cases, immediate deployment of dengue rt pcr test kits can quickly confirm dengue etiology, identify the predominant serotype, and enable targeted public health interventions. This capability saves lives, reduces healthcare burden, and prevents wider community transmission by informing vector control measures effectively.
Industry Trends, Market Dynamics, and Manufacturer Comparison
Evolving Landscape of Dengue Diagnostics
The market for dengue pcr test kits is experiencing robust growth driven by increasing disease incidence, greater awareness, and technological advancements. Key trends include:
- Automation and Integration: A significant shift towards fully automated or semi-automated platforms that integrate nucleic acid extraction, amplification, and detection, reducing hands-on time and improving throughput, especially for high-volume laboratories.
- Point-of-Care (POC) PCR: Development of simpler, rapid, and portable PCR systems suitable for near-patient testing or resource-limited settings. While still evolving, POC PCR aims to bring the precision of molecular diagnostics closer to the patient.
- Multiplexing Capabilities: Kits capable of simultaneously detecting dengue virus and other co-circulating arboviruses (e.g., Zika, Chikungunya) in a single reaction, streamlining differential diagnosis.
- Next-Generation Sequencing (NGS) Integration: While not a primary diagnostic tool, NGS is increasingly used in conjunction with PCR for advanced epidemiological studies, tracking viral evolution, and identifying novel DENV strains.
- Enhanced Data Management: Software solutions for seamless data integration, analysis, and reporting, supporting better public health surveillance.
Key Parameters for Dengue PCR Test Kits - Comparative Table
When evaluating dengue pcr test kits, laboratories and procurement specialists consider several critical parameters to ensure optimal performance, cost-effectiveness, and compatibility with existing infrastructure. Below is a comparative table of typical parameters found across leading dengue rt pcr test products on the market, illustrating key differentiators:
| Parameter | Typical Range / Feature | Implication for Users |
|---|---|---|
| Detection Method | Real-Time RT-PCR (qPCR) | Quantitative results, faster TAT, closed system (less contamination risk). |
| Target Genes | NS5, C/prM, E regions (highly conserved) | Ensures broad detection across all serotypes and viral strains. |
| Serotype Differentiation | DENV 1, 2, 3, 4 (multiplex or singleplex) | Crucial for epidemiological surveillance and understanding outbreak dynamics. |
| Sensitivity (LoD) | 5-20 copies/reaction or 10-50 PFU/mL | Ability to detect very low viral loads, enabling early diagnosis. |
| Specificity | >99.5% (no cross-reactivity with common respiratory/arboviruses) | Minimizes false positives, crucial for accurate diagnosis. |
| Turnaround Time (TAT) | 2-4 hours (from sample to result, excluding extraction) | Faster patient management and public health response. |
| Sample Volume Input | 100-200 µL serum/plasma | Standardized input for consistent performance. |
| Required Instrumentation | Real-Time PCR cycler (e.g., ABI 7500, Bio-Rad CFX96), nucleic acid extractor (manual/automated) | Compatibility with existing lab equipment. |
| Kit Format | Single-tube, pre-aliquoted, or lyophilized reagents | Ease of use, reduced pipetting errors, storage stability. |
| Regulatory Approvals | CE-IVD, FDA EUA/510(k), WHO Prequalification | Ensures quality, safety, and performance for clinical use. |
| Shelf Life | 12-24 months at specified storage conditions | Inventory management and cost efficiency. |
Leading manufacturers in this space often provide comprehensive solutions, from nucleic acid extraction reagents and automated platforms to validated dengue rna pcr test kits and accompanying software. Factors influencing choice include validation status for different sample types, ease of workflow integration, cost per test, and the level of technical support provided.
Custom Solutions, Case Studies, and Partnering for Excellence
Tailoring Diagnostic Capabilities to Specific Needs
While off-the-shelf dengue pcr test kits cater to a broad range of diagnostic needs, many laboratories and public health systems require customized solutions to address unique challenges, such as specific regional serotype prevalence, integration with existing laboratory information management systems (LIMS), or specialized research requirements.
- Custom Panel Development: Manufacturers can collaborate to develop multiplex dengue rt pcr test panels that include not only DENV serotypes but also other co-circulating arboviruses prevalent in a specific geographical area, optimizing diagnostic workflows.
- Automation Integration: For high-throughput labs, customized automation solutions can be engineered, involving robotic liquid handlers and integrated nucleic acid extraction platforms that seamlessly interface with dengue rna pcr test protocols, drastically increasing daily sample processing capacity.
- Software Customization: Tailored software solutions for data analysis, result reporting, and epidemiological tracking can be developed to meet specific reporting formats or integrate with public health surveillance databases.
- Bulk Reagent Supply & OEM: For large-scale testing programs or other diagnostic kit manufacturers, suppliers often offer bulk quantities of core reagents (primers, probes, enzymes) or Original Equipment Manufacturer (OEM) solutions, allowing for internal kit assembly and cost optimization.
Illustrative Case Study: Enhancing Public Health Surveillance in an Endemic Region
A large public health laboratory in Southeast Asia faced challenges in rapidly identifying dengue serotypes during outbreaks, leading to delays in targeted public health interventions. Their existing serological methods provided delayed information, often after the peak of viral transmission.
Solution: The laboratory partnered with a leading diagnostic solutions provider to implement a high-throughput dengue pcr test system. This involved:
- Deployment of Automated Extraction: Installation of an automated nucleic acid extraction platform capable of processing 96 samples in under an hour, significantly reducing manual labor and improving consistency.
- Implementation of a Multiplex RT-qPCR Kit: Adoption of a CE-IVD marked dengue rt pcr test kit designed to simultaneously detect and differentiate all four DENV serotypes. This kit was validated specifically for their local prevalent strains.
- Comprehensive Training and Support: The provider offered extensive on-site training for laboratory personnel on instrument operation, assay execution, data analysis, and troubleshooting. Ongoing technical support and regular calibration services were also part of the agreement.
Outcome: Within six months of implementation, the laboratory reported a 70% reduction in turnaround time for dengue confirmation and serotype identification. This enabled public health officials to identify emerging serotypes faster, leading to more targeted vector control campaigns and better resource allocation. The increased capacity also allowed for broader surveillance, enhancing the overall understanding of dengue epidemiology in the region.
Ensuring Trust and Authority: Quality, Support, and Certifications
In the sensitive field of medical diagnostics, especially for critical tests like the dengue pcr test, trust, authority, and unwavering quality are non-negotiable. Reputable manufacturers adhere to the highest international standards, ensuring product reliability and customer satisfaction.
Commitment to Quality and Regulatory Compliance
Manufacturers of dengue rna pcr test kits operate under strict quality management systems (QMS). Key certifications and compliance standards include:
- ISO 13485:2016 Certification: This international standard specifies requirements for a comprehensive quality management system for the design and manufacture of medical devices. It ensures that all processes, from raw material sourcing to final product release, are rigorously controlled and documented.
- CE-IVD Marking: Products intended for in vitro diagnostic use within the European Union must bear the CE-IVD mark, indicating compliance with the European In Vitro Diagnostic Medical Devices Regulation (IVDR).
- FDA Clearance/EUA: For the United States market, products undergo rigorous review by the U.S. Food and Drug Administration (FDA) to obtain 510(k) clearance or Emergency Use Authorization (EUA) during public health emergencies, ensuring safety and efficacy.
- WHO Prequalification: For global procurement and deployment, particularly in low- and middle-income countries, WHO prequalification provides an additional layer of assurance regarding quality, safety, and performance of diagnostic products.
Adherence to these standards guarantees that every dengue pcr test kit delivered is manufactured under controlled conditions, precisely formulated, and performs consistently as per its specifications. This commitment to quality minimizes diagnostic errors and contributes directly to patient safety and public health.
Customer Support and Service Offerings
Beyond product quality, the level of customer support is a critical determinant of long-term partnerships in the B2B diagnostics sector. Comprehensive support typically includes:
- Technical Support: Access to expert technical teams for troubleshooting, assay optimization, and protocol guidance via phone, email, or online portals.
- On-site Training: Hands-on training for laboratory staff on instrument operation, assay setup, and data analysis, ensuring proficient use of dengue pcr test systems.
- Service & Maintenance: Regular instrument calibration, preventive maintenance, and rapid response for repairs minimize downtime and ensure continuous operation.
- Application Support: Guidance on specific research applications or adapting assays for unique clinical scenarios.
Delivery Cycle: For standard orders, typical delivery cycles for dengue pcr test kits range from 2-4 weeks, depending on region and inventory. Urgent orders or high-volume procurements may require specific logistical planning, often facilitated by dedicated account managers. Global distribution networks ensure timely and secure delivery of temperature-sensitive reagents.
Warranty and Return Policy: Standard warranties typically cover manufacturing defects for the stated shelf life of the kit when stored and used as per instructions. Manufacturers usually have clear return policies for defective products, ensuring client satisfaction and confidence.
Frequently Asked Questions (FAQ) about Dengue PCR Testing
- Q: What is the ideal sample type and collection time for a dengue pcr test?
A: Serum or plasma collected during the acute febrile phase (ideally within 0-7 days of symptom onset) is most suitable for dengue RNA PCR test due to peak viremia during this period.
- Q: Can a dengue pcr test differentiate between primary and secondary dengue infections?
A: A dengue pcr test primarily detects the presence of the virus and can identify the serotype. Differentiation between primary and secondary infections typically relies on serological markers (IgM/IgG ratios) or a combination of molecular and serological results.
- Q: How does the dengue rt pcr test compare to NS1 antigen tests?
A: Both are useful for early diagnosis. PCR generally offers higher sensitivity and specificity, especially for detecting low viral loads, and can differentiate serotypes. NS1 antigen tests are rapid but can have variable sensitivity depending on the serotype and viral load.
- Q: Is dengue pcr test prone to false negatives due to viral mutations?
A: Reputable dengue pcr test kits are designed with primers and probes targeting highly conserved regions of the viral genome to minimize the impact of mutations. Regular surveillance and kit updates are also part of manufacturers' quality assurance.
- Q: What are the biosafety requirements for performing a dengue pcr test?
A: Work with clinical samples should be conducted in a BSL-2 (Biosafety Level 2) laboratory with appropriate personal protective equipment (PPE) and sterile techniques to prevent contamination and exposure.
Conclusion: The Future of Dengue Diagnostics
The dengue pcr test has firmly established itself as an indispensable tool in the global fight against dengue fever. Its ability to provide rapid, highly sensitive, and specific detection of viral RNA, along with serotype identification, empowers clinicians to make informed decisions for patient care and enables public health agencies to effectively monitor and control outbreaks. As technological advancements continue, we can anticipate even more integrated, automated, and potentially point-of-care solutions that will further enhance accessibility and efficiency of dengue rt pcr test and dengue rna pcr test methodologies worldwide. Investing in robust PCR diagnostic capabilities is not merely an operational choice; it is a strategic imperative for global health security and effective disease management.
References and Further Reading
- World Health Organization. Dengue and severe dengue. Available from: https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue
- Gubler, D. J. (2012). The global pandemic of dengue/dengue hemorrhagic fever: Current status and prospects for control. Journal of clinical virology, 54(4), 293-301.
- Peeling, R. W., Artsob, M. H., & Chua, A. R. (2010). Diagnosis of Dengue Infection. Clinical Infectious Diseases, 51(suppl_4), S338-S342.
- Lanciotti, R. S., et al. (1992). Rapid detection and typing of dengue viruses from clinical samples by using reverse transcriptase-polymerase chain reaction. Journal of Clinical Microbiology, 30(3), 545-551.
- CDC Dengue Clinical Guidance. Available from: https://www.cdc.gov/dengue/healthcare-providers/index.html
- TDR/WHO. Diagnostics for Dengue. Available from: https://tdr.who.int/disease-areas/dengue/diagnostics-for-dengue
- Centers for Disease Control and Prevention. Arboviral Diseases Branch. Dengue Virus. Available from: https://www.cdc.gov/ncezid/dvbd/diseases/dengue/index.html
Related PRODUCTS
-
Rapid BK Virus PCR: Accurate & Quantitative Diagnostics
NewsAug.29,2025 -
HLA B27 PCR Test Kit: Accurate & Rapid Detection
NewsAug.28,2025 -
Accurate Yellow Fever PCR Test Kits | Fast & Reliable Results
NewsAug.27,2025 -
Expert Respiratory Panel Lab Services for Rapid Diagnosis
NewsAug.26,2025 -
Accurate Legionella Pneumophila Detection PCR Kits
NewsAug.19,2025