أغسطس . 29, 2025 05:20 Back to list
Rapid BK Virus PCR: Accurate & Quantitative Diagnostics
Industry Trends in BK Virus Detection and Monitoring
The landscape of post-transplant care has been significantly reshaped by advancements in molecular diagnostics, particularly concerning opportunistic viral infections. Among these, the BK polyomavirus (BKV) presents a substantial challenge to transplant recipients, often leading to nephropathy (BKVN) in kidney transplant patients or hemorrhagic cystitis (BKHCV) in hematopoietic stem cell transplant (HSCT) recipients. Accurate and timely detection of BKV is paramount for mitigating these severe complications, making reliable bk virus pcr a cornerstone of contemporary clinical management.
Recent trends indicate a growing demand for highly sensitive, specific, and quantitative assays. The shift towards personalized medicine necessitates precise viral load monitoring to guide immunosuppression adjustments, thereby balancing graft protection with infection control. Furthermore, multiplex PCR platforms are gaining traction, allowing for the simultaneous detection of multiple pathogens or the co-quantification of BKV and other polyomaviruses like JC virus (JCV), enhancing diagnostic efficiency. The integration of automation and high-throughput capabilities in laboratory settings is also a key trend, driven by the increasing volume of transplant procedures globally and the need for rapid turnaround times.
The global market for molecular diagnostics, particularly in infectious disease management, continues its robust expansion. Projections suggest a sustained compound annual growth rate (CAGR) exceeding 8% for molecular diagnostics over the next five years, fueled by technological innovations, rising incidence of infectious diseases, and an aging population requiring more complex medical interventions. Within this growth, advanced quantitative PCR solutions for viral pathogens like BKV are critical components, addressing a direct clinical need for precision and reliability.
Technical Specifications and Product Deep Dive: Cowingene BK Virus And JC Virus Detection Kit
The Cowingene BK Virus And JC Virus Detection Kit exemplifies the forefront of molecular diagnostics, offering a comprehensive solution for the quantitative detection of BK virus (BKV) and JC virus (JCV) DNA. This kit is designed to provide robust and reliable results, critical for managing immunocompromised patients. The methodology employed is real-time fluorescence PCR, a gold standard for viral load quantification.
Detailed Process Flow: Manufacturing and Application
The manufacturing process for a sophisticated diagnostic kit like the Cowingene BK Virus And JC Virus Detection Kit adheres to stringent quality control and industry standards, ensuring product integrity and performance.
- Raw Material Sourcing & QC: High-grade oligonucleotides (primers and probes), enzymes (e.g., Taq polymerase), deoxynucleotide triphosphates (dNTPs), and buffer components are sourced from validated suppliers. Each batch undergoes rigorous incoming quality control to verify purity, concentration, and absence of contaminants (e.g., nucleases, non-specific DNA).
- Reagent Formulation & Mixing: Proprietary formulations of master mixes are prepared under controlled cleanroom conditions. This involves precise volumetric dispensing and thorough mixing of enzyme cocktails, specific primers and probes for BKV and JCV targets, and an internal control for monitoring DNA extraction and PCR inhibition.
- Component Dispensing & Filling: Automated, high-precision dispensing systems fill individual reaction tubes or multi-well plates with the formulated reagents. This process is conducted in a sterile environment to prevent contamination and ensure consistent reaction volumes across all units.
- Lyophilization (Optional but Recommended for Stability): Depending on the product variant, reagents may undergo lyophilization (freeze-drying). This process removes water, significantly extending the shelf life and stability of the reagents at ambient temperatures, reducing cold chain requirements during transport and storage.
- Assembly, Labeling & Packaging: The filled and potentially lyophilized components are assembled into kits, including controls and instruction manuals. Each kit is meticulously labeled with batch numbers, expiration dates, and CE-IVD/NMPA markings. Packaging is designed to protect reagents from physical damage and environmental factors (light, humidity) during transit and storage.
- Final Product Quality Control & Release: Extensive performance testing is conducted on representative samples from each manufacturing lot. This includes sensitivity (Limit of Detection - LoD), specificity, linearity, precision, and reproducibility tests. These tests are benchmarked against ISO 13485 and national regulatory standards (e.g., NMPA in China, CE-IVD in Europe), ensuring the kit meets its performance claims before release.
For the end-user, the application process for Cowingene's kit typically follows these steps:
- Sample Collection: Collection of patient samples, typically plasma, urine, or cerebrospinal fluid (CSF).
- Nucleic Acid Extraction: Isolation of viral DNA from the collected samples using validated commercial extraction kits or automated systems. An internal control is usually co-extracted to monitor the efficiency of this step.
- PCR Setup: Preparation of the reaction mixture by combining extracted DNA, Cowingene's master mix, and positive/negative controls.
- Real-Time PCR Amplification & Detection: Running the prepared reactions on a compatible real-time PCR instrument. The instrument monitors fluorescence signals emitted by probes as target DNA amplifies, allowing for quantitative analysis of viral load.
- Data Analysis & Interpretation: Using specialized software to analyze amplification curves and generate quantitative results, expressed as copies/mL.
Product Materials and Standards
The kit's components are manufactured using medical-grade plastics and highly purified biochemical reagents. Manufacturing processes adhere strictly to ISO 13485:2016 for medical devices. The kit is CE-IVD marked, signifying compliance with European In Vitro Diagnostic Device Regulations, and is registered with relevant national authorities. The shelf life of the kit is typically 12-18 months when stored at recommended temperatures (-20°C).
Adapted Advantages for Diagnostic Kits
- Enhanced Efficiency & Throughput: While not "energy saving" in a traditional sense, the kit's design for rapid and accurate quantification significantly saves laboratory resources, reduces manual intervention, and accelerates diagnostic turnaround times, analogous to operational energy efficiency.
- Reagent Stability & Robustness: Rather than "corrosion resistance," the kit is formulated for exceptional reagent stability and robustness, ensuring consistent performance across varying laboratory conditions within its specified storage parameters. This minimizes batch-to-batch variability and provides reliable results even under challenging clinical sample matrices.
Cowingene BK Virus And JC Virus Detection Kit Specifications
| Parameter | Specification |
|---|---|
| Product Name | Cowingene BK Virus And JC Virus Detection Kit |
| Target Viruses | BK Polyomavirus (BKV), JC Polyomavirus (JCV) |
| Detection Method | Real-Time Fluorescence Quantitative PCR (qPCR) |
| Target Genes | Specific regions of BKV VP1 gene and JCV VP1 gene |
| Internal Control | Present, for monitoring DNA extraction and PCR inhibition |
| Sample Types | Plasma, Urine, CSF (extracted DNA) |
| Limit of Detection (LoD) | BKV: ≤ 200 copies/mL; JCV: ≤ 200 copies/mL (Varies slightly by instrument) |
| Linear Range | 1.0 x 10^3 to 1.0 x 10^9 copies/mL |
| Specificity | ≥ 99% (No cross-reactivity with common respiratory/enteric pathogens or other polyomaviruses) |
| Accuracy | CV ≤ 5% (Intra-assay), CV ≤ 8% (Inter-assay) |
| Storage Conditions | -20°C ± 5°C |
| Shelf Life | 12 months |
| Compatible Instruments | Mainstream real-time PCR systems (e.g., Applied Biosystems 7500, Bio-Rad CFX96) |
Application Scenarios for BK Virus Quantitative PCR
The utility of bk virus quantitative pcr extends across various critical clinical settings, primarily driven by the need to monitor and manage viral reactivation in immunocompromised individuals. Its precision allows for proactive intervention, significantly improving patient outcomes.
- Renal Transplant Recipients: This is the most prominent application. BKV infection can lead to BKV-associated nephropathy (BKVN), a significant cause of graft dysfunction and loss. Regular monitoring of BKV viral load in plasma and urine allows clinicians to identify patients at risk, predict BKVN onset, and guide reduction of immunosuppressive therapy to prevent graft rejection while controlling viral replication.
- Hematopoietic Stem Cell Transplant (HSCT) Recipients: BKV reactivation in HSCT patients is often associated with hemorrhagic cystitis (BKHCV), a painful and debilitating condition. Quantitative PCR helps in early detection and management, distinguishing between viral shedding and active disease, and informing antiviral treatment strategies.
- Other Solid Organ Transplant (SOT) Recipients: While less common than in kidney transplants, BKV can affect other SOT recipients, including heart, liver, and lung transplant patients, often presenting with varied clinical manifestations. Surveillance with bk virus pcr contributes to comprehensive post-transplant care.
- HIV-infected Individuals: Although less frequent, BKV nephropathy can also occur in HIV-positive patients, especially those with advanced immunosuppression. bk pcr offers a valuable tool for diagnosis and monitoring in this specific population.
- Research and Clinical Trials: Beyond routine diagnostics, reliable quantitative PCR assays are essential for research into BKV pathogenesis, evaluating new antiviral therapies, and understanding immune responses in transplant patients.

Figure 1: Conceptual illustration of a BK virus PCR diagnostic workflow in a clinical lab.
Technical Advantages of Cowingene's Solution
The Cowingene BK Virus And JC Virus Detection Kit offers distinct technical advantages that position it as a premier choice for clinical laboratories and research institutions:
- High Sensitivity and Specificity: Utilizing optimized primer and probe designs, the kit achieves an impressively low Limit of Detection (LoD), enabling early detection of viral reactivation even at very low copy numbers. This is critical for pre-emptive intervention. Its high specificity ensures accurate identification of BKV and JCV without cross-reactivity from other common pathogens or host DNA.
- Quantitative Accuracy and Wide Linear Range: The kit provides precise viral load quantification across a broad linear range (1.0 x 10^3 to 1.0 x 10^9 copies/mL), allowing for effective monitoring of disease progression and response to therapy. This quantitative accuracy is essential for making informed clinical decisions regarding immunosuppression adjustment.
- Multiplex Detection Capability: The ability to simultaneously detect and quantify both BKV and JCV in a single reaction saves time, reagents, and sample volume, streamlining laboratory workflow and reducing costs. This is particularly valuable in settings where co-infection or differential diagnosis is relevant.
- Robust Internal Control: The inclusion of an internal control monitors the entire process, from nucleic acid extraction to PCR amplification, mitigating false-negative results due to extraction failures or PCR inhibition. This enhances the trustworthiness and reliability of every test result.
- Standardized and Reproducible Results: Manufactured under ISO 13485 quality management systems, the kit ensures batch-to-batch consistency and high reproducibility of results, crucial for longitudinal patient monitoring and multi-center studies.
- Compatibility with Mainstream Instruments: Designed for compatibility with various widely available real-time PCR platforms, the kit can be readily integrated into existing laboratory infrastructure, minimizing the need for new equipment investment.
Vendor Comparison: Cowingene vs. Competitors
When evaluating diagnostic kits for bk virus pcr, laboratories consider several critical factors beyond just price. Performance metrics, ease of use, regulatory compliance, and support are paramount. The Cowingene BK Virus And JC Virus Detection Kit stands out in a competitive market.
| Feature/Parameter | Cowingene Kit | Competitor A (e.g., Brand X) | Competitor B (e.g., Brand Y) |
|---|---|---|---|
| Detection Method | Real-Time Fluorescence qPCR | Real-Time Fluorescence qPCR | Real-Time Fluorescence qPCR |
| Target Viruses | BKV & JCV (Multiplex) | BKV only (or separate kits) | BKV & JCV (Multiplex) |
| Limit of Detection (LoD) | ≤ 200 copies/mL (BKV/JCV) | 250-500 copies/mL | ≤ 200 copies/mL |
| Linear Range | 1.0 x 10^3 - 1.0 x 10^9 copies/mL | 5.0 x 10^2 - 1.0 x 10^8 copies/mL | 1.0 x 10^3 - 1.0 x 10^8 copies/mL |
| Internal Control | Yes (co-extracted, co-amplified) | Yes (amplification only) | Yes (co-extracted, co-amplified) |
| Regulatory Approvals | CE-IVD, NMPA | CE-IVD, FDA (specific regions) | CE-IVD, local approvals |
| Automation Compatibility | High (open platform) | Medium (some proprietary systems) | High (open platform) |
| Customer Support | Dedicated technical support, training | Standard support | Online resources |
Cowingene's kit differentiates itself with its optimized multiplex design for simultaneous BKV and JCV detection, coupled with a robust internal control that monitors both extraction and amplification efficiency. While competitors offer solid products, Cowingene often provides a more cost-effective solution without compromising on the high standards of sensitivity, specificity, and quantitative accuracy required for critical clinical diagnostics. Our commitment to comprehensive technical support and flexible automation compatibility further enhances our value proposition for laboratories seeking reliable bk virus quantitative pcr solutions.
Customized Solutions and Application Case Studies
Customized Solutions
Cowingene understands that laboratories have unique requirements based on their sample throughput, existing equipment, and specific research or clinical needs. We offer flexible, customized solutions for our bk pcr products, including:
- Bulk Packaging Options: For high-throughput laboratories, we provide reagents in larger volumes or specialized multi-well plate formats to optimize workflow and reduce per-test costs.
- Instrument Validation Support: While our kit is designed for broad compatibility, we offer extensive technical support for validation on specific real-time PCR instruments or automated extraction systems not explicitly listed, ensuring seamless integration into your lab's existing setup.
- Custom Panel Development: For research institutions or specialized clinical centers, we can explore custom assay configurations, such as incorporating additional viral targets or specific polyomavirus genotypes, leveraging our molecular biology expertise.
- Training and Implementation: We provide comprehensive training programs for laboratory personnel, ensuring proficient use of the kit and optimal performance within your specific operational environment.
Application Case Studies
These case studies highlight the practical impact and reliability of Cowingene's BK Virus And JC Virus Detection Kit in real-world clinical scenarios.
Case Study 1: Early Detection Preventing Renal Allograft Loss
A major transplant center in Europe adopted Cowingene's multiplex bk virus pcr kit for routine post-kidney transplant surveillance. Patient X, a 45-year-old male, showed a gradual increase in plasma BKV viral load from 1,500 copies/mL to 15,000 copies/mL over three weeks, identified by the Cowingene kit's quantitative precision. This sub-clinical elevation, well below thresholds for overt BKVN symptoms, prompted an early reduction in immunosuppression. Subsequent monitoring showed a steady decline in BKV viral load, preventing progression to BKVN and preserving graft function. The high sensitivity and reliability of the kit allowed for timely intervention, averting potential graft rejection and the need for re-transplantation.
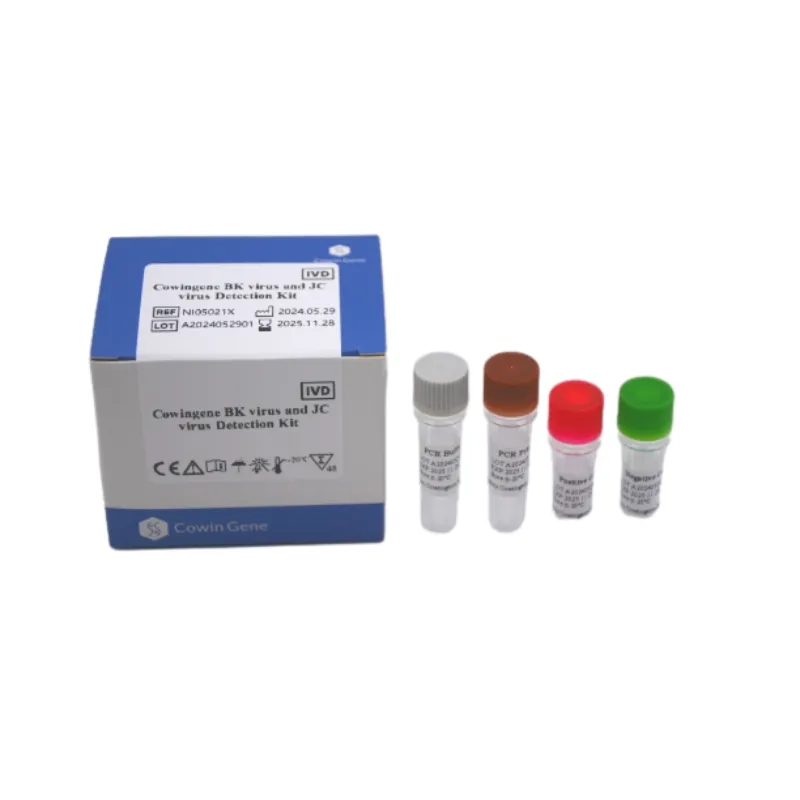
Figure 2: Representative real-time PCR amplification curves for BK virus detection.
Case Study 2: Differential Diagnosis in HSCT-Associated Hemorrhagic Cystitis
At a prominent oncology hospital, Patient Y, a 28-year-old female post-HSCT, presented with severe hemorrhagic cystitis. Initial differential diagnoses included BKV, adenovirus, and cytomegalovirus. Using Cowingene's kit, a high urinary BKV viral load (>1.0 x 10^7 copies/mL) was rapidly detected, while JCV and other viral loads were negligible. This precise identification of BKV as the causative agent allowed clinicians to initiate targeted antiviral therapy and adjust fluid management, leading to significant symptom improvement and resolution of hemorrhagic cystitis within two weeks. The multiplexing capability of the kit was instrumental in quickly narrowing down the etiology, saving critical diagnostic time.
These cases underscore Cowingene's commitment to providing diagnostic tools that directly contribute to better patient management and improved clinical outcomes through accurate and timely viral quantification.
Trustworthiness: FAQ, Lead Time, Warranty, and Support
Frequently Asked Questions (FAQ) about BK Virus PCR
Q: What is the primary purpose of bk virus pcr testing?
A: The primary purpose is to quantitatively detect and monitor BK polyomavirus (BKV) DNA in patient samples, mainly plasma and urine, particularly in immunocompromised individuals such as organ transplant recipients, to diagnose and manage BKV-associated nephropathy or hemorrhagic cystitis.
Q: How does Cowingene's kit handle potential PCR inhibition?
A: Our kit includes a robust internal control that is co-extracted and co-amplified with the target DNA. This internal control monitors both the efficiency of nucleic acid extraction and the presence of PCR inhibitors, ensuring the reliability of negative results and accurate quantification.
Q: Is the Cowingene BK Virus And JC Virus Detection Kit compatible with automated extraction systems?
A: Yes, the kit is compatible with DNA extracted using most commercially available automated nucleic acid extraction systems. We provide protocols and support for validating the kit with your specific automated platform.
Q: What is the difference between qualitative and quantitative BKV PCR?
A: Qualitative bk virus pcr simply detects the presence or absence of BKV DNA. Quantitative PCR, like the Cowingene kit, measures the precise amount (viral load) of BKV DNA. Quantitative results are crucial for monitoring disease progression, assessing response to treatment, and making clinical decisions, as viral load changes often precede clinical symptoms.
Lead Time and Fulfillment
Cowingene maintains robust inventory and efficient logistics to ensure timely delivery of our products. Standard lead time for the BK Virus And JC Virus Detection Kit is typically 2-4 business days for domestic orders and 7-14 business days for international shipments, following order confirmation. Expedited shipping options are available upon request. All kits are shipped under controlled temperature conditions (-20°C with dry ice) to maintain product integrity.
Warranty Commitments
Cowingene warrants that the BK Virus And JC Virus Detection Kit will perform according to the specifications provided in the product insert for its stated shelf life when stored and used as directed. Should any product defect or failure to meet specifications be identified, Cowingene commits to replacing the product or issuing a credit, subject to our standard warranty terms and conditions. Our commitment is to ensure your laboratory receives reliable, high-performing diagnostic tools.
Customer Support and After-Sales Service
Our dedicated technical support team comprises experienced molecular biologists and application specialists ready to assist with product inquiries, assay optimization, troubleshooting, and data interpretation. Support is available via phone, email, and live chat during business hours. We also offer on-site training and technical visits for larger installations or complex integration needs, ensuring that our partners maximize the utility and performance of Cowingene products.
References
- Hirsch, H.H., & Randhawa, P. (2019). BK Polyomavirus in Solid Organ Transplantation. American Journal of Transplantation, 19(Suppl 2), 244-252.
- Vanchiere, J.A., & Cron, R.Q. (2014). Polyomavirus-Associated Nephropathy. Pediatric Nephrology, 29(1), 19-27.
- Bohl, D.L., & Brennan, D.C. (2007). BK virus nephropathy and its impact on renal transplantation. Clinical Journal of the American Society of Nephrology, 2(Suppl 1), S36-S42.
- Tong, C.Y.W., et al. (2010). Review of the diagnosis and management of BK virus infection in kidney transplantation. Journal of Clinical Virology, 49(Suppl 1), S48-S54.
- Google Scholar and PubMed Search: "BK virus quantitative PCR performance" and "JC virus quantitative PCR sensitivity" for general performance benchmarks of molecular diagnostic kits.
This is the first article
Related PRODUCTS
-
HLA B27 PCR Test Kit: Accurate & Rapid Detection
NewsAug.28,2025 -
Accurate Yellow Fever PCR Test Kits | Fast & Reliable Results
NewsAug.27,2025 -
Expert Respiratory Panel Lab Services for Rapid Diagnosis
NewsAug.26,2025 -
Fast & Accurate Dengue PCR Test for Early Diagnosis
NewsAug.21,2025 -
Accurate Legionella Pneumophila Detection PCR Kits
NewsAug.19,2025