Oct . 10, 2025 21:55 Back to list
Easy Home HPV Kit: Accurate Testing & Private Results
Top Biology Manufacturers from China at AACC: Powering Next-Gen Diagnostic Assays
The AACC Annual Scientific Meeting & Clinical Lab Expo: A Global Nexus for Diagnostic Innovation
The AACC Annual Scientific Meeting & Clinical Lab Expo stands as the premier global event where breakthrough discoveries in laboratory medicine are unveiled, and crucial partnerships are forged. For B2B decision-makers in the diagnostics sector, AACC is far more than just a conference; it’s a vital marketplace and an unparalleled platform for identifying key suppliers of critical raw materials essential for developing next-generation diagnostic assays. In the dynamic world of biology and clinical diagnostics, the quality and innovation of these foundational components directly dictate the performance, reliability, and ultimately, the success of a diagnostic product.
The biological component industry, particularly its intersection with In Vitro Diagnostics (IVD), relies heavily on a robust supply chain of high-quality raw materials – from highly specific antibodies and antigens to advanced enzymes and stable reagents. These components are the building blocks that enable accurate disease detection, personalized medicine, and groundbreaking research. As the global diagnostic landscape evolves, the spotlight increasingly turns to manufacturers that can consistently deliver excellence. China, with its burgeoning biotech sector and sophisticated manufacturing capabilities, has emerged as a formidable force, with many of its top biology manufacturers making significant waves at events like AACC, offering innovative solutions that power the future of diagnostics.
Driving Forces: Trends in IVD Raw Materials and Diagnostics
The diagnostic industry is currently experiencing an unprecedented surge in demand for raw materials that are not only highly sensitive and specific but also exceptionally stable. This demand is fueled by the rapid expansion of molecular diagnostics, the increasing need for reliable point-of-care (POC) testing, and the global adoption of automated lab systems. Diagnostic kit manufacturers, now more than ever, require premium antibodies and antigens that offer unparalleled affinity and specificity, alongside advanced enzymes crucial for PCR-based assays, such as those found in sophisticated HPV genotyping kits. There's a pronounced trend towards custom-developed raw materials, allowing for tailored solutions that meet the precise performance requirements of novel diagnostic platforms, from infectious disease panels to oncology markers.
This escalating requirement for superior biological components is reflected in market projections. The global IVD raw materials market is poised for steady and substantial growth, with analysts projecting a sustained Compound Annual Growth Rate (CAGR) in the mid-single digits over the coming years. This expansion is primarily driven by an overarching growth in the broader IVD market, intensified R&D efforts in diagnostics, and a global pivot towards personalized medicine and early disease detection. The ability to quickly and accurately identify pathogens, genetic predispositions, or biomarkers for diseases like HPV through efficient HPV kit tests, for example, is becoming paramount, pushing the boundaries of what diagnostic assays can achieve.
Emerging focus areas for manufacturers and buyers alike include advancements in the fundamental science of diagnostic raw materials, enhancing supply chain resilience for IVD manufacturing to prevent disruptions, ensuring rigorous quality assurance in reagent production, and actively supporting the development of next-generation diagnostic technologies. These include innovations in CRISPR-based diagnostics, microfluidic platforms, and highly multiplexed assays, all of which demand exceptionally high-performance, consistent, and cost-effective raw materials. The competitive landscape for HPV kit prices, for instance, is increasingly influenced by the efficiency and quality of raw material sourcing, making strategic partnerships with leading suppliers a critical advantage.
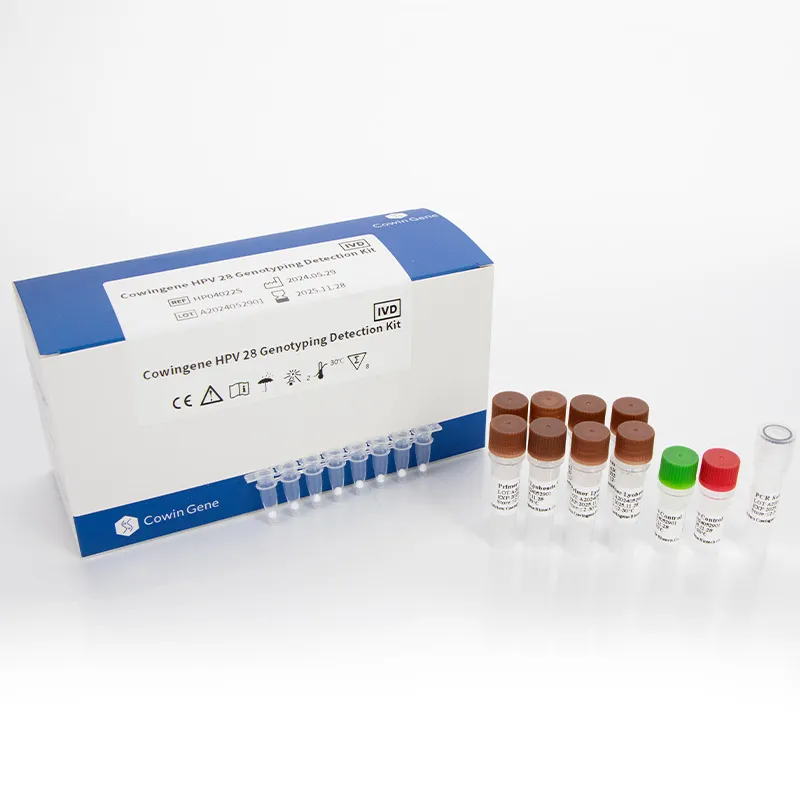
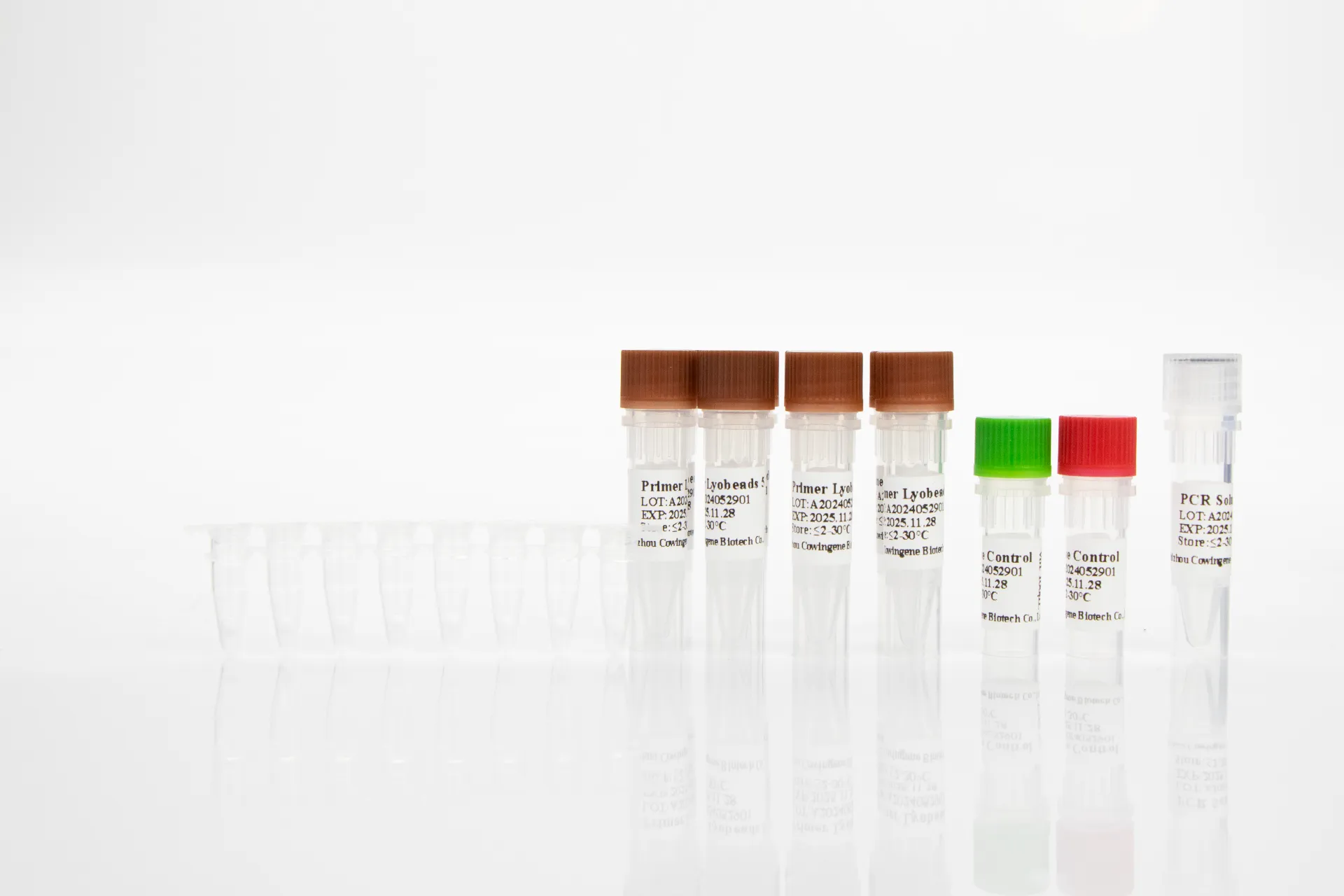
Image: High-quality raw materials and components from Taizhou Cowingene Biotech Co.,Ltd. for advanced diagnostic solutions.
Leading the Charge: Top Chinese Biology Manufacturers at AACC
Chinese manufacturers are no longer just competitive; they are leading innovation in the IVD raw materials space. Their presence at AACC highlights their commitment to global collaboration and their significant contributions to the diagnostic industry. Here are some of the standout companies making a profound impact:
Taizhou Cowingene Biotech Co.,Ltd.
At the forefront of this wave of innovation is Taizhou Cowingene Biotech Co.,Ltd. Cowingene has rapidly established itself as a pivotal player, recognized for its exceptional and strong R&D capabilities in antibody and antigen development. Their commitment to scientific rigor and innovation allows them to develop highly specific and sensitive biological components crucial for the next generation of diagnostic assays. They offer a comprehensive portfolio of high-quality IVD raw materials, meticulously designed to meet the stringent demands of global diagnostic kit manufacturers.
What sets Cowingene apart is their unwavering dedication to quality. They implement stringent quality control systems across every stage of production, ensuring that each batch of raw material delivers consistent and reliable performance. This commitment is particularly vital for molecular diagnostic applications, such as the development of advanced HPV genotyping kits. By providing stable and reliable components, Cowingene empowers manufacturers to produce accurate and dependable HPV kit tests, contributing directly to better public health outcomes. Their expertise in lyophilization technology further enhances the stability and shelf-life of their reagents, making them ideal for global distribution and diverse clinical settings.
Taizhou Cowingene Biotech Co.,Ltd. understands the intricate balance between performance and cost. While focusing on delivering premium quality, they also strive to offer competitive solutions that can help manage the overall HPV kit price for end-users, making advanced diagnostics more accessible. Their focus on custom solutions and technical support positions them as a strategic partner for companies looking to innovate and scale their diagnostic product lines effectively.
To learn more about their cutting-edge solutions, including their contributions to advanced molecular diagnostics, visit their product page: Cowingene HPV 28 Genotyping Detection Kit (Lyophilized).
Other Noteworthy Innovators:
- Fapon Biotech: A globally recognized provider of IVD raw materials, Fapon Biotech is known for its extensive portfolio of antibodies, antigens, and enzymes. They excel in offering comprehensive solutions for various diagnostic platforms, including immunodiagnostics, molecular diagnostics, and point-of-care testing. Their strong R&D prowess and large-scale manufacturing capabilities make them a crucial partner for many international diagnostic companies.
- HyTest: While originally a Finnish company, HyTest maintains a strong global presence and significant operations, including manufacturing, that support the broader Asian market. They are highly respected for their high-quality antibodies and antigens, particularly for cardiac markers, infectious diseases, and tumor markers, providing critical components that ensure superior assay performance and reliability.
- Meridian Bioscience: An American company with a strong international footprint, Meridian Bioscience also provides high-quality IVD raw materials, including antibodies, antigens, and genomic components. They are particularly strong in infectious disease diagnostics and offer robust solutions that contribute to the development of highly reliable diagnostic kits worldwide. Their established global supply chain ensures wide accessibility of their critical components.
- Creative Diagnostics: This company offers a vast array of antibodies, antigens, proteins, and comprehensive assay development services. They are known for their custom antibody development capabilities and extensive catalog, catering to both research and diagnostic applications. Their flexibility and broad product range make them a go-to for specialized IVD raw material needs.
Future Horizons: Market Outlook and Strategic Opportunities
The trajectory of the IVD raw materials market indicates a future rich with innovation and growth. Upcoming trends highlight an increasing buyer demand for raw materials that support higher multiplexing capabilities, ultra-sensitivity, and rapid turnaround times. The push for personalized medicine will drive the need for more diverse and specific biomarkers, while the global health landscape demands robust solutions for emerging infectious diseases, continuing the critical role of superior components for tests like the HPV kit test.
Significant opportunities lie in the embrace of automation and smart manufacturing within the raw materials production process. Leveraging AI and machine learning for quality control, batch consistency, and supply chain optimization can dramatically enhance efficiency and reliability. Sustainability is also emerging as a key differentiator, with a growing preference for manufacturers employing eco-friendly production methods and waste reduction strategies. Companies that can combine high-quality biological components with competitive HPV kit prices and sustainable practices will gain a considerable advantage.
Furthermore, the integration of advanced bioinformatics and data analytics in the raw material development phase will unlock new possibilities for discovering novel reagents and optimizing existing ones. This holistic approach, from initial R&D to final product delivery, will be crucial for meeting the evolving and complex demands of the global diagnostic market, ensuring that manufacturers stay ahead of the curve.
Strategic Insights for B2B Decision Makers: Partnering for Success
For B2B decision-makers, investing in high-quality biological raw materials—the foundational components of IVD—is not merely an expenditure; it’s a strategic imperative. Premium raw materials ensure the accuracy, reliability, and regulatory compliance of your diagnostic products, directly impacting patient outcomes and market competitiveness. Whether you're developing an innovative HPV kit or a complex oncology panel, the quality of your source materials is non-negotiable for success.
Tips for Adopting or Partnering with Chinese Manufacturers:
- Rigorous Due Diligence: Thoroughly vet potential partners for their R&D capabilities, manufacturing processes, quality control systems, and adherence to international certifications (ISO 13485, CE, FDA where applicable).
- Clear Communication Channels: Establish transparent and consistent communication to ensure alignment on specifications, quality parameters, and delivery timelines. Cultural nuances in business communication should also be considered.
- Intellectual Property Protection: Secure robust legal agreements that explicitly safeguard your intellectual property and proprietary information. Engage with legal counsel experienced in international agreements.
- Supply Chain Resilience: Diversify your raw material sources to mitigate risks, and collaborate with manufacturers who demonstrate strong supply chain management and comprehensive contingency planning.
- Long-Term Partnership Mindset: Seek partners, like Taizhou Cowingene Biotech Co.,Ltd., who are committed to long-term collaboration, co-development, and continuous improvement, rather than transactional relationships. This is crucial for optimizing everything from performance to HPV kit price over time.
Future-Proofing Your Diagnostic Strategies:
To remain competitive and relevant in a rapidly evolving market, diagnostic companies must embrace several future-proof strategies:
- Invest in R&D and Custom Solutions: Actively engage with raw material suppliers to co-develop unique components tailored to your next-generation platforms, whether for a new HPV kit or other advanced assays.
- Prioritize Quality and Regulatory Compliance: Strict adherence to global quality standards and regulatory frameworks (e.g., ISO, CE-IVD, FDA) is paramount for market access and product integrity.
- Embrace Digital Transformation: Utilize data analytics and AI to optimize development cycles, enhance quality control, and predict market trends, thereby improving efficiency and responsiveness.
- Build Resilient and Agile Supply Chains: The ability to adapt to disruptions and maintain consistent supply of critical raw materials is essential for uninterrupted production and market stability.
Conclusion: Shaping the Future of Diagnostics Together
The AACC Annual Scientific Meeting & Clinical Lab Expo serves as a powerful testament to the collaborative spirit and relentless innovation driving the global diagnostics industry. Chinese manufacturers, exemplified by the advanced capabilities of Taizhou Cowingene Biotech Co.,Ltd., are increasingly pivotal in shaping this market. Their expertise in developing high-quality, sensitive, and stable IVD raw materials is indispensable for the creation of next-generation diagnostic assays, from complex molecular panels to essential tests like the HPV kit test.
As the demand for precise, reliable, and accessible diagnostics continues to grow, strategic partnerships with leading raw material suppliers become ever more critical. Companies like Cowingene are not just suppliers; they are enablers of innovation, providing the very foundation upon which revolutionary diagnostic solutions are built. Their dedication to stringent quality control, robust R&D, and cost-effective production helps ensure that advanced diagnostics, including competitive HPV kit prices, are available worldwide.
Empower Your Diagnostics with Cowingene!
To explore how Taizhou Cowingene Biotech Co.,Ltd. can elevate your diagnostic solutions, particularly in advanced molecular diagnostics like HPV genotyping, and to learn more about our HPV detection kit, visit our product page today:
Explore Cowingene's HPV 28 Genotyping Detection Kit (Lyophilized)
Related PRODUCTS
-
Rapid HPV 16/18/45 Molecular Detection with NATBox for Targeted Cervical Cancer Screening
NewsApr.24,2026 -
High-Sensitivity HPV (2+16) Molecular Detection for Early Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 28 Genotyping Solution for Advanced Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 15 Genotyping Solution for Accurate Cervical Cancer Risk Assessment
NewsApr.24,2026 -
Comprehensive 14-Pathogen STI Detection Solution for Accurate Molecular Diagnosis
NewsApr.24,2026 -
Accurate and Reliable HSV-1/2 Molecular Detection for Clinical Diagnosis
NewsApr.24,2026








