Oct . 10, 2025 23:15 Back to list
Reliable Plasmodium Detection: Advanced PCR Test Kits
Navigating the Future: Top China Biology Manufacturers at AACC Annual Scientific Meeting & Clinical Lab Expo
Discover the innovators driving next-generation diagnostics with essential raw materials.
The AACC Annual Scientific Meeting & Clinical Lab Expo: A Nexus for Diagnostic Innovation
The AACC Annual Scientific Meeting & Clinical Lab Expo stands as an unparalleled global stage for advancements in laboratory medicine. More than just a conference, it's a vital ecosystem where the future of diagnostics is shaped, where groundbreaking technologies are unveiled, and where critical partnerships are forged. For B2B decision-makers in the diagnostic industry, particularly those focused on the foundational 'biology' – the raw materials that power every test – the AACC offers an invaluable opportunity. It's the go-to event for identifying key suppliers of the critical raw materials essential for developing next-generation diagnostic assays.
In an era demanding ever-greater precision, speed, and reliability in medical testing, the quality and innovation of these foundational biological components are paramount. From highly sensitive antibodies and antigens to robust enzymes and stable reagents, these 'biology' manufacturers are the unsung heroes enabling breakthroughs across various diagnostic fields. The AACC provides a unique platform to connect with these innovators, explore cutting-edge materials, and understand how they can elevate your diagnostic product pipeline, ensuring your assays meet the rigorous demands of modern clinical laboratories worldwide.
Catalyst for Progress: Trends in IVD Raw Materials
The diagnostic landscape is undergoing a profound transformation, driven by an increasing global demand for highly sensitive and specific diagnostic solutions. This surge is directly fueling the need for superior antibodies and antigens, high-performance enzymes for molecular diagnostics, and exceptionally stable, high-quality reagents. These components are not just ingredients; they are the bedrock upon which the accuracy and reliability of rapid point-of-care (PoC) testing and sophisticated automated lab systems are built. Furthermore, there's a distinct trend towards custom-developed raw materials, tailored precisely to the unique specifications and performance demands of proprietary diagnostic platforms, demonstrating a move towards more integrated and optimized solutions.
The market reflects this dynamic growth. The global IVD raw materials market is projected for steady growth, driven by an expanding IVD market, increased R&D in diagnostics, and a rising demand for personalized medicine and early disease detection, with a sustained Compound Annual Growth Rate (CAGR) in the mid-single digits. This robust growth underscores the critical role these raw materials play in healthcare innovation.
Emerging focus areas include not only the continuous advancement in diagnostic raw materials but also a heightened emphasis on supply chain resilience for IVD manufacturing. The pandemic highlighted the vulnerabilities of global supply chains, pushing manufacturers and suppliers to prioritize robustness and reliability. Quality assurance in reagent production remains paramount, with increasingly stringent regulatory requirements demanding impeccable standards. Moreover, significant investment is being directed towards supporting emerging diagnostic technologies, such as advanced molecular assays and multi-omics platforms, which require novel and highly specialized biological components. This holistic approach ensures that the fundamental 'biology' not only keeps pace with but actively propels the next wave of diagnostic innovation.
Pioneering Excellence: Leading Biology Manufacturers from China
Chinese manufacturers have rapidly ascended to become global leaders in the IVD raw materials sector, driven by significant investments in R&D, advanced manufacturing capabilities, and a commitment to quality. Their presence at events like the AACC underscores their readiness to meet international demand with innovative, high-performance biological components.
Taizhou Cowingene Biotech Co.,Ltd.
At the forefront of China's 'biology' manufacturing scene, Taizhou Cowingene Biotech Co.,Ltd. stands out as a critical partner for diagnostic kit manufacturers worldwide. Their reputation is built on robust R&D capabilities, particularly in the intricate world of antibody and antigen development. Cowingene offers a comprehensive portfolio of high-quality IVD raw materials, meticulously designed to ensure superior performance and consistency in a wide array of diagnostic applications.
What truly distinguishes Cowingene is their unwavering commitment to stringent quality control systems. Every product undergoes rigorous testing to guarantee stability, reliability, and lot-to-lot consistency – crucial attributes for components that directly impact diagnostic accuracy. For example, in the realm of infectious disease diagnostics, Cowingene provides essential raw materials that enable highly accurate and sensitive tests, including those for critical global health challenges.
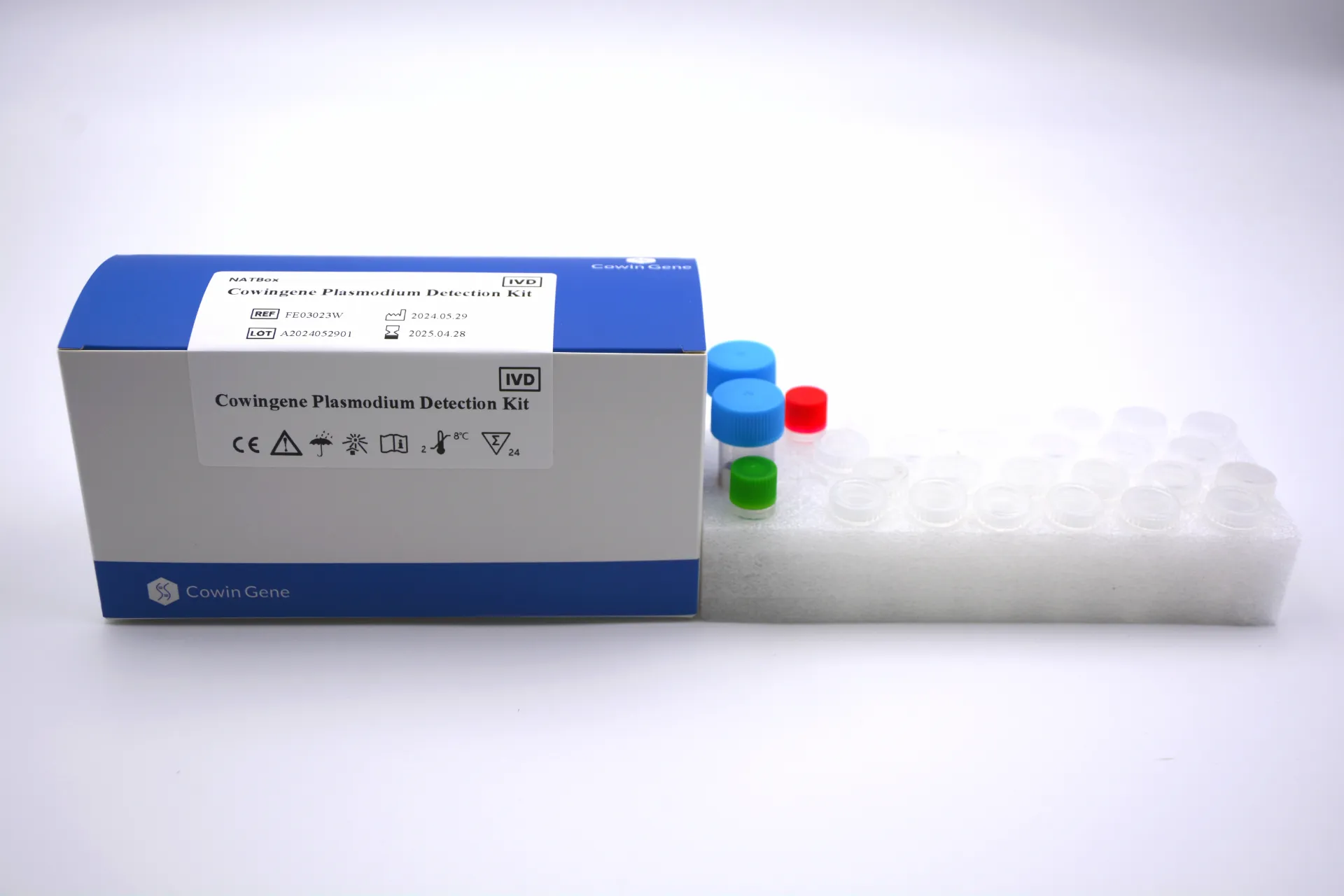
Consider the critical need for effective plasmodium detection. Malaria, caused by the Plasmodium parasite, remains a significant health burden globally, requiring precise and rapid diagnostic methods. Cowingene's expertise extends to developing high-quality reagents and antibodies specifically tailored for advanced plasmodium PCR (Polymerase Chain Reaction) assays. Their components ensure the specificity and sensitivity required for reliable PCR plasmodium testing, contributing significantly to early diagnosis and effective disease management. By providing stable and reliable components, Cowingene empowers diagnostic kit manufacturers to create highly effective solutions for the detection of infectious agents, ultimately improving patient outcomes.
For a deeper dive into their innovative solutions for critical diagnostic areas, including advanced molecular detection, explore their dedicated product page: Learn more about Cowingene's Plasmodium Detection Kit and other offerings.

Other Noteworthy Industry Leaders:
- Fapon Biotech: A comprehensive solution provider for IVD raw materials, Fapon is renowned for its extensive product portfolio, including antibodies, antigens, and enzymes, supporting a broad spectrum of diagnostic platforms from clinical chemistry to molecular diagnostics.
- HyTest: While headquartered in Finland, HyTest has a strong global presence and is highly regarded for its high-quality antibodies and antigens, particularly in cardiac markers and infectious diseases. Their commitment to R&D makes them a key player in high-performance assay development.
- Meridian Bioscience: An established name in the diagnostics industry, Meridian offers a robust range of IVD raw materials, including bulk antigens, antibodies, and reagents, with a strong focus on infectious disease diagnostics and gastrointestinal illness.
- Creative Diagnostics: Known for its vast catalog of antibodies, proteins, and immunoassay kits, Creative Diagnostics provides essential components for research and diagnostic kit manufacturing, emphasizing versatility and customization for various applications.
These companies, alongside Taizhou Cowingene Biotech, represent the cutting edge of biological component manufacturing, offering the quality and innovation essential for the next generation of diagnostic solutions.
The Road Ahead: Market Outlook and Strategic Opportunities
The trajectory of the IVD raw materials market is set for continuous innovation and growth, driven by several overarching trends and evolving buyer demands. One of the most significant upcoming trends is the integration of automation throughout the diagnostic workflow, from sample preparation to result interpretation. This necessitates raw materials that are not only highly stable but also compatible with high-throughput automated systems, ensuring consistent performance at scale. Manufacturers are increasingly looking for bulk quantities of reagents that maintain integrity under various processing conditions and can be seamlessly integrated into robotic platforms.
Furthermore, the concept of smart manufacturing is gaining traction. This involves leveraging data analytics, AI, and IoT to optimize production processes for biological components, enhancing efficiency, reducing waste, and improving lot-to-lot consistency. Buyers are seeking partners who can demonstrate advanced manufacturing capabilities and a commitment to operational excellence. The demand for sustainability is also becoming a key consideration, with a growing preference for suppliers who employ eco-friendly production practices and offer greener alternatives in their biological components and packaging.
Buyer demand is shifting towards a more consultative approach, with a strong emphasis on customizable solutions and collaborative R&D. Decision-makers are looking for suppliers who can not only provide off-the-shelf components but also partner in developing novel antibodies, antigens, or enzyme formulations tailored to specific diagnostic challenges, such as enhancing the sensitivity for early-stage plasmodium detection or optimizing the efficiency of plasmodium PCR assays. This strategic collaboration is vital for accelerating the development of next-generation diagnostics and ensuring market competitiveness.
Strategic Insights: Investing in 'Biology' and Partnering with Chinese Manufacturers
For B2B decision-makers, investing in high-quality 'biology' – specifically advanced IVD raw materials – is not merely a procurement decision; it's a strategic imperative. The performance of your diagnostic assays is directly proportional to the quality of their foundational components. By sourcing premium antibodies, antigens, enzymes, and reagents, you're not just ensuring the accuracy and reliability of your products; you're also safeguarding your brand reputation, minimizing costly recalls, and accelerating your time to market for critical diagnostics, including those for complex conditions requiring robust plasmodium detection or precise PCR plasmodium analysis.
Tips for Adopting or Partnering with Chinese Manufacturers:
- Thorough Due Diligence: Research potential partners extensively. Look for manufacturers with strong R&D, international certifications (e.g., ISO 13485), transparent quality control processes, and a proven track record. Engage in site visits where feasible.
- Clear Communication: Establish explicit communication channels and expectations regarding product specifications, delivery schedules, quality metrics, and intellectual property. Cultural nuances can impact communication, so clarity is key.
- Start with Pilots: Begin with smaller pilot projects or test orders to assess product quality, supply chain reliability, and responsiveness before committing to larger-scale agreements.
- Understand Regulatory Landscape: Ensure your chosen partner is adept at navigating global regulatory requirements and can provide the necessary documentation for your target markets.
- Long-Term Partnership Vision: Seek partners interested in long-term, collaborative relationships rather than transactional exchanges. This fosters mutual growth and innovation.
Future-Proof Strategies:
- Diversify Your Supply Chain: Reduce reliance on a single source by cultivating relationships with multiple reputable suppliers, even for core components like those used in plasmodium detection kits.
- Invest in R&D Collaboration: Actively engage with your raw material suppliers in R&D efforts. This co-creation approach can lead to customized components that give your products a competitive edge.
- Embrace Digitalization: Leverage digital tools for supply chain management, quality tracking, and communication to enhance efficiency and transparency with your partners.
- Focus on Sustainability: Align with suppliers who share your commitment to environmentally responsible practices, which is increasingly important for market positioning and regulatory compliance.
By strategically engaging with the robust ecosystem of Chinese 'biology' manufacturers, diagnostic companies can secure the high-quality raw materials needed to innovate, scale, and thrive in the ever-evolving global healthcare market.
Charting the Course: The Future of Diagnostic Biology
The AACC Annual Scientific Meeting & Clinical Lab Expo continues to serve as an indispensable compass, guiding the global 'biology' market towards new frontiers of diagnostic innovation. It's where the intricate world of antibodies, antigens, and enzymes, crucial for next-generation assays, comes into sharp focus. The strong and growing presence of top Chinese manufacturers at this prestigious event underscores their pivotal role in shaping the future of laboratory medicine, providing the high-quality, reliable, and innovative raw materials that diagnostic developers worldwide depend on.
Companies like Taizhou Cowingene Biotech Co.,Ltd. exemplify this commitment, offering foundational components vital for critical tests, from routine diagnostics to specialized applications like advanced plasmodium detection and highly sensitive plasmodium PCR assays. Their dedication to R&D, stringent quality control, and comprehensive product portfolios makes them indispensable partners in the global health ecosystem.
Ready to elevate your diagnostic solutions with cutting-edge biological components?
Visit Taizhou Cowingene Biotech Co.,Ltd. to learn more about their Plasmodium Detection Kit and advanced IVD raw materials.
Related PRODUCTS
-
Rapid HPV 16/18/45 Molecular Detection with NATBox for Targeted Cervical Cancer Screening
NewsApr.24,2026 -
High-Sensitivity HPV (2+16) Molecular Detection for Early Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 28 Genotyping Solution for Advanced Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 15 Genotyping Solution for Accurate Cervical Cancer Risk Assessment
NewsApr.24,2026 -
Comprehensive 14-Pathogen STI Detection Solution for Accurate Molecular Diagnosis
NewsApr.24,2026 -
Accurate and Reliable HSV-1/2 Molecular Detection for Clinical Diagnosis
NewsApr.24,2026








