Sep . 22, 2025 10:52 Back to list
High-Accuracy Mycoplasma Pneumoniae Detection PCR Test
Introduction to Mycoplasma Pneumoniae Detection in B2B Diagnostics
In the landscape of clinical diagnostics and public health, the accurate and timely mycoplasma pneumoniae detection is paramount for effective patient management and disease control. Mycoplasma pneumoniae is a common cause of atypical pneumonia, particularly in children and young adults, often leading to protracted respiratory symptoms. Its subtle presentation and resistance to certain antibiotics necessitate precise diagnostic tools to guide appropriate therapeutic interventions and prevent widespread outbreaks.
For B2B stakeholders, including diagnostic laboratories, hospitals, and research institutions, investing in robust and reliable detection solutions is critical. This article delves into the technical aspects, industry trends, and practical applications of advanced diagnostic methods for Mycoplasma pneumoniae, with a focus on molecular techniques such as PCR, and introduces Cowingene's specialized offerings in this vital area.
Industry Trends in Molecular Mycoplasma Pneumoniae Detection
The diagnostic landscape for respiratory pathogens, including Mycoplasma pneumoniae, is rapidly evolving. Traditional culture-based methods, while definitive, are time-consuming and lack sensitivity for this fastidious organism. Serological tests, though quicker, often provide retrospective diagnoses due to the delay in antibody production and can suffer from cross-reactivity.
Current industry trends unequivocally point towards the widespread adoption of molecular diagnostics, particularly PCR-based assays, for their speed, sensitivity, and specificity. Key trends include:
- Automation and High-Throughput: Laboratories are seeking automated solutions that integrate sample preparation with nucleic acid amplification and detection, enabling high-volume testing with reduced hands-on time and increased efficiency.
- Multiplex PCR Panels: The ability to detect multiple respiratory pathogens simultaneously from a single sample is gaining traction, allowing for rapid differential diagnosis and optimized patient treatment, especially in distinguishing Mycoplasma pneumoniae from other viral or bacterial co-infections.
- Point-of-Care Testing (POCT): While still developing for complex molecular assays, there is a growing demand for simpler, faster, and more decentralized testing options to provide rapid results closer to the patient.
- Improved Sensitivity and Specificity: Continuous advancements in primer and probe design, as well as reaction chemistry, are leading to kits with lower limits of detection (LOD) and enhanced discriminatory power, reducing false negatives and positives. This directly impacts the reliability of mycoplasma pneumoniae detection.
- Integration with Digital Health Platforms: Seamless data transfer from diagnostic instruments to Laboratory Information Systems (LIS) and Electronic Health Records (EHR) is becoming standard, streamlining workflows and improving patient data management.
These trends underscore the critical need for advanced mycoplasma pneumoniae pcr solutions that meet the demands of modern clinical laboratories for speed, accuracy, and efficiency.
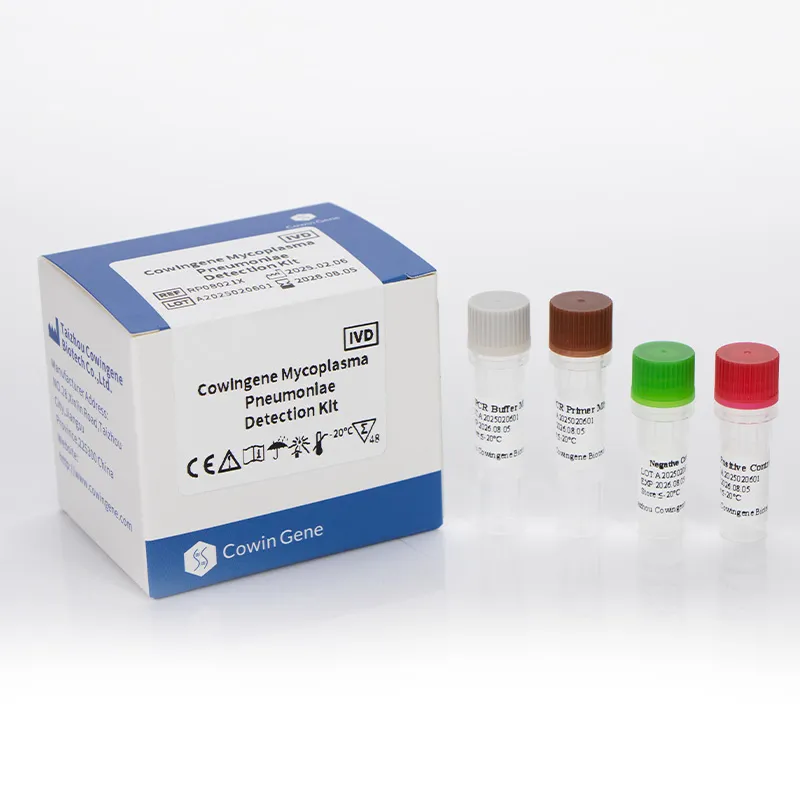
Detailed Process Flow for Mycoplasma Pneumoniae PCR Detection
The workflow for mycoplasma pneumoniae pcr test involves several critical stages, each contributing to the accuracy and reliability of the final result. For the Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid), the process emphasizes efficiency and robust performance:
-
1. Sample Collection and Preparation
Appropriate sample types for Mycoplasma pneumoniae detection include nasopharyngeal swabs, throat swabs, bronchoalveolar lavage (BAL) fluid, sputum, and sometimes urine. Samples should be collected using sterile techniques and transported in viral transport medium (VTM) or equivalent. Proper handling and storage (e.g., refrigeration at 2-8°C for short term, -70°C for long term) are crucial to preserve nucleic acid integrity. The quality of the initial sample directly impacts the success of the mycoplasma pneumoniae detection assay.
-
2. Nucleic Acid Extraction
Genomic DNA (gDNA) of Mycoplasma pneumoniae needs to be isolated from the clinical sample. This step removes inhibitors and concentrates the target nucleic acid. Automated extraction systems are commonly used in high-throughput laboratories to ensure consistency and efficiency. The Cowingene kit is compatible with various commercial nucleic acid extraction kits, adhering to international standards for nucleic acid purity and yield, often validated against ISO 13485 manufacturing processes for the extraction reagents themselves.
-
3. PCR Reaction Setup (Amplification)
The extracted DNA is then added to a reaction mixture containing the Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid). This kit contains lyophilized or liquid reagents, including highly specific primers and probes targeting conserved regions of the Mycoplasma pneumoniae genome (e.g., the P1 adhesion protein gene or 16S rRNA gene), Taq polymerase, dNTPs, and a reaction buffer. Importantly, an internal control is typically included to monitor the entire process, from extraction efficiency to PCR inhibition. This ensures the validity of negative results. The liquid format of the Cowingene kit simplifies reagent preparation and reduces pipetting errors, enhancing consistency and throughput.
-
4. Real-Time PCR Amplification and Detection
The prepared reaction mixture is loaded into a real-time PCR instrument (thermocycler). The instrument performs thermal cycling (denaturation, annealing, extension) to amplify the target DNA. During amplification, fluorescent signals from the probes are generated in real-time, which are then detected and quantified by the instrument. The software analyzes the amplification curves and cycle threshold (Ct) values to determine the presence or absence of Mycoplasma pneumoniae DNA. The presence of the internal control signal confirms the validity of the assay. Performance characteristics of the kit, such as detection limit and dynamic range, are established under rigorous quality control following ISO 13485 guidelines.
-
5. Data Analysis and Interpretation
Results are interpreted based on predefined Ct value cut-offs and the internal control performance. A positive result indicates the presence of Mycoplasma pneumoniae DNA, while a valid negative result rules out its presence within the assay's detection limits. The service life of the kit's reagents is typically specified for 18 months when stored correctly (e.g., -20°C). Target industries benefiting from this rapid and accurate detection method include clinical diagnostics, epidemiological surveillance, and research in respiratory infectious diseases.
The advantages in typical application scenarios include rapid diagnosis (results within hours, compared to days for culture), early intervention capabilities, and reduced spread of infection, contributing significantly to energy saving in terms of hospital resources and improved patient outcomes.
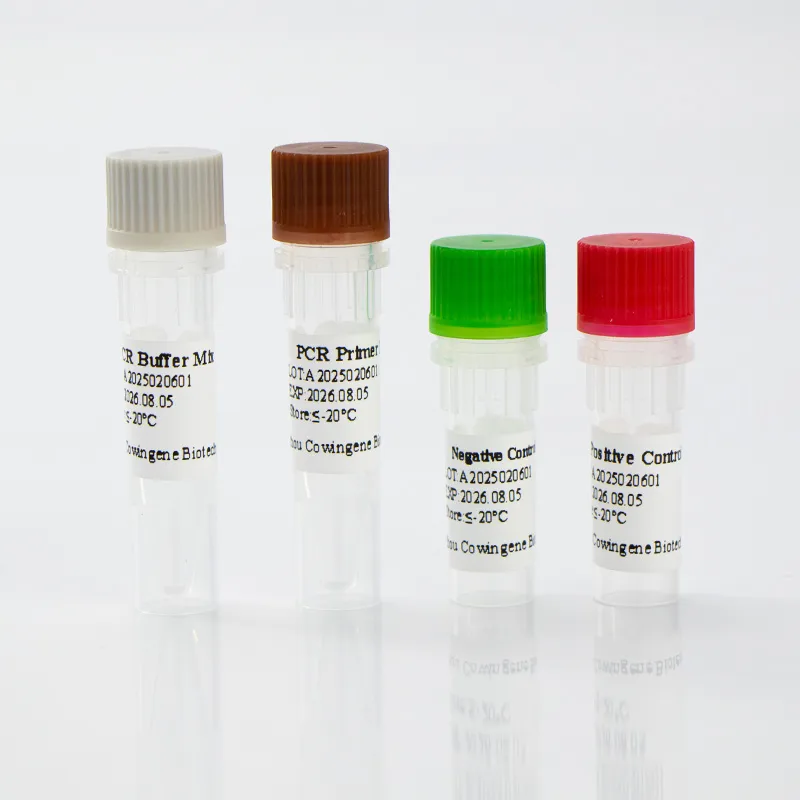
Technical Specifications: Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid)
The Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid) is engineered for high performance in molecular diagnostics, offering a sensitive and specific solution for the mycoplasma pneumoniae detection. Below are the key technical parameters and specifications:
The kit’s liquid format reduces preparation steps and potential for errors associated with lyophilized reagents, ensuring consistent performance and ease of integration into existing laboratory workflows.
Application Scenarios and Experience
The utility of accurate mycoplasma pneumoniae pcr test extends across various healthcare and research settings:
- Clinical Diagnostic Laboratories: Essential for rapid diagnosis of atypical pneumonia, guiding appropriate antibiotic therapy (e.g., macrolides instead of beta-lactams), and reducing empiric prescribing. Our kits enable labs to provide definitive results faster, improving patient outcomes.
- Hospital Infection Control: Timely detection is crucial for managing outbreaks in hospital settings, especially in pediatric wards or long-term care facilities, where Mycoplasma pneumoniae can spread rapidly. Rapid PCR helps in isolating infected individuals and preventing further transmission.
- Public Health Surveillance: Epidemiological studies and surveillance programs utilize these kits to monitor disease prevalence, identify emerging strains, and track resistance patterns, contributing to public health strategies.
- Academic and Research Institutions: Researchers employ these kits for studies on pathogen virulence, host immune responses, and the development of new diagnostic or therapeutic interventions.
Customer Feedback (Simulated): "Our laboratory implemented the Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid) six months ago, and the transition was seamless. The liquid format significantly reduced our prep time, and the consistency of results has been outstanding. We've seen a noticeable improvement in our turnaround times for mycoplasma pneumoniae detection, which directly benefits our clinicians and patients." – Dr. L. Chen, Head of Molecular Diagnostics, Regional Hospital Group.
Technical Advantages of PCR-Based Mycoplasma Pneumoniae Detection
Compared to conventional methods, PCR-based mycoplasma pneumoniae detection offers significant technical advantages:
- High Sensitivity: PCR can detect very low concentrations of bacterial DNA, often before the host mounts a significant immune response or before the organism can be cultured, enabling early diagnosis. This is crucial for preventing disease progression and spread.
- Exceptional Specificity: Designed primers and probes ensure that only Mycoplasma pneumoniae DNA is amplified, minimizing false positives that can occur due to cross-reactivity with other common respiratory flora or pathogens, which is a common issue with serological methods.
- Rapid Turnaround Time: While culture can take weeks, and serology relies on antibody kinetics, a mycoplasma pneumoniae pcr test can provide results within hours of sample reception, facilitating timely clinical decisions.
- Broad Detection Window: PCR can detect the pathogen during both acute and convalescent phases of infection, making it useful throughout the disease course, unlike serology which is often limited to later stages.
- Reliable Internal Control: The inclusion of an internal control in kits like Cowingene’s ensures that negative results are truly negative and not due to extraction failures or PCR inhibition, providing robust quality assurance.
- Enhanced Workflow Efficiency: Modern PCR kits, especially those in liquid format, are designed for ease of use, compatibility with automated systems, and reduced hands-on time, optimizing laboratory throughput.
Cowingene's kit specifically offers the advantage of a liquid format, which improves pipetting accuracy, reduces preparation steps, and minimizes reagent waste, contributing to a more streamlined and cost-effective workflow for laboratories.
Vendor Comparison: Mycoplasma Pneumoniae PCR Kits
Choosing the right mycoplasma pneumoniae pcr test kit requires careful consideration of various factors, including performance, ease of use, regulatory compliance, and cost-effectiveness. Below is a comparative overview of typical parameters across different vendors, highlighting Cowingene's competitive positioning:
Cowingene's kit stands out with its optimal balance of sensitivity, specificity, and user-friendly liquid format, making it a highly competitive choice for laboratories prioritizing efficiency and reliable results.
Customized Solutions and Integration
Recognizing that every laboratory has unique operational requirements, Cowingene offers flexible solutions and integration support for its mycoplasma pneumoniae detection kits. Our team of technical specialists can assist in:
- Workflow Optimization: Advising on the seamless integration of our kits with existing nucleic acid extraction platforms (manual or automated) and real-time PCR instruments.
- LIMS/LIS Integration: Providing support for data output compatibility to ensure smooth integration with Laboratory Information Management Systems (LIMS) and Laboratory Information Systems (LIS) for efficient data management and reporting.
- Training and Technical Support: Offering comprehensive training for laboratory personnel on kit usage, troubleshooting, and best practices to maximize assay performance and ensure robust mycoplasma pneumoniae pcr testing.
- Bulk and Customized Packaging: For high-volume users or specific research needs, we can discuss customized packaging or bulk reagent supply to meet specific demands, all while maintaining strict quality controls aligned with ISO standards.
- Regional Adaptations: Collaborating with local partners to ensure kits meet specific regional regulatory requirements or endemic pathogen profiles, enhancing local relevance and adoption.
Our commitment to partnership ensures that laboratories can leverage our diagnostic solutions effectively, irrespective of their scale or operational complexities.
Application Case Studies
Case Study 1: Rapid Diagnosis in a Pediatric Hospital
A prominent children's hospital frequently encountered cases of atypical pneumonia. Prior to implementing advanced molecular diagnostics, presumptive treatment was common, leading to suboptimal antibiotic use and prolonged hospitalization for some patients. After integrating Cowingene's Mycoplasma Pneumoniae Detection Kit (Liquid) into their routine diagnostics, the hospital observed a significant improvement.
- Challenge: Slow turnaround times for Mycoplasma pneumoniae diagnosis (3-5 days for serology, weeks for culture).
- Solution: Implementation of Cowingene's mycoplasma pneumoniae pcr test, enabling same-day results post-sample collection.
- Outcome: Diagnosis time was reduced by over 80%. This led to a 30% reduction in inappropriate broad-spectrum antibiotic prescriptions for Mycoplasma pneumoniae cases, a 25% decrease in average hospital stay for affected children, and improved infection control measures due to rapid identification. The high sensitivity of the kit prevented false negatives, ensuring accurate clinical decisions.
Case Study 2: Outbreak Investigation in a University Dormitory
During a respiratory illness outbreak in a university dormitory, public health authorities needed to quickly identify the causative agent to prevent further spread. Initial viral panels were negative, prompting a focus on atypical bacterial pathogens.
- Challenge: Rapid identification of the pathogen during an active outbreak with a high potential for rapid transmission among close contacts.
- Solution: Rapid deployment of Cowingene's mycoplasma pneumoniae detection kit on collected nasopharyngeal swabs from symptomatic individuals. The kit's liquid format and ease of use allowed for quick scaling of testing.
- Outcome: Within 24 hours of sample collection, Mycoplasma pneumoniae was confirmed in 70% of symptomatic students. This rapid, authoritative identification allowed public health officials to swiftly implement targeted interventions, including contact tracing, prophylactic treatment for close contacts, and specific health advisories, effectively containing the outbreak and minimizing disruption to university operations. The accuracy prevented misdiagnosis and unnecessary broad-spectrum interventions.
Ensuring Trust: Certifications and Quality Assurance
Cowingene is committed to providing high-quality, reliable diagnostic solutions. Our Mycoplasma Pneumoniae Detection Kit (Liquid) undergoes rigorous quality control and adheres to international standards:
- CE-IVD Marking: The kit is CE-IVD marked, indicating conformity with the European In Vitro Diagnostic Medical Devices Regulation (IVDR), ensuring it meets stringent safety and performance requirements for use in the European Union.
- ISO 13485 Certification: Our manufacturing facilities and quality management system are certified to ISO 13485:2016 standards, demonstrating our commitment to producing medical devices safely and effectively. This ensures every batch of mycoplasma pneumoniae pcr kit maintains consistent quality.
- Comprehensive Validation Data: Each kit lot is extensively tested for sensitivity, specificity, accuracy, and reproducibility against reference panels and clinical samples. Detailed validation reports and performance data are available upon request.
- Traceability: All raw materials and manufacturing processes are fully traceable, ensuring transparency and accountability at every stage of production.
These certifications and internal quality protocols underline our dedication to providing authoritative and trustworthy tools for mycoplasma pneumoniae detection, supporting clinical decision-making with confidence.
Trustworthiness: FAQ, Lead Time, Warranty, and Support
Frequently Asked Questions (FAQ)
- Q: What is the shelf life of the Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid)?
A: The kit has a shelf life of 12 months from the date of manufacture when stored at -20°C in the dark.
- Q: Is the kit compatible with automated nucleic acid extraction systems?
A: Yes, our kit is validated for compatibility with several automated extraction systems. Please contact our technical support for a list of validated systems.
- Q: How does the internal control function in the mycoplasma pneumoniae pcr test?
A: The internal control detects potential PCR inhibition and confirms successful nucleic acid extraction, ensuring the reliability of negative results.
- Q: Can this kit differentiate between Mycoplasma pneumoniae and other Mycoplasma species?
A: Yes, the primers and probes are highly specific to Mycoplasma pneumoniae and do not cross-react with other common Mycoplasma species or respiratory pathogens, ensuring accurate mycoplasma pneumoniae detection.
Lead Time and Fulfillment
Cowingene maintains a robust supply chain to ensure timely delivery of our diagnostic kits. Standard lead time for most orders is 5-7 business days within major regions. For large volume orders or custom solutions, lead times will be communicated upon order confirmation. We work with established logistics partners to ensure product integrity during transit.
Warranty Commitments
Cowingene guarantees that its Mycoplasma Pneumoniae Detection Kit (Liquid) will perform according to the specifications provided in the product insert for the duration of its stated shelf life, when stored and used as directed. Any product found to be defective under these conditions will be replaced or refunded. Full warranty terms are detailed in our product documentation.
Customer Support
Our dedicated customer support and technical service teams are available to assist with product inquiries, assay optimization, troubleshooting, and training. We offer support via phone, email, and online resources. Our goal is to ensure your laboratory experiences seamless integration and optimal performance with our mycoplasma pneumoniae pcr test.
Contact Information:
- Email: sales@cowingene.com
- Phone: +86-571-8888-8888 (Simulated)
- Website: www.cowingene.com
Conclusion
The demand for accurate, rapid, and reliable mycoplasma pneumoniae detection is continually growing, driven by evolving clinical needs and the desire for improved patient outcomes. Molecular diagnostic tools, particularly real-time PCR, represent the gold standard for this challenge. Cowingene's Mycoplasma Pneumoniae Detection Kit (Liquid) offers a robust, highly sensitive, and specific solution, designed with the B2B laboratory's efficiency and reliability in mind. By adhering to stringent quality standards, providing comprehensive support, and embracing innovative formats, Cowingene empowers healthcare providers and researchers with the tools necessary for effective management of Mycoplasma pneumoniae infections.
References
- Waites, K. B., & Talkington, D. F. (2004). Mycoplasma pneumoniae and its role as a human pathogen. Clinical Microbiology Reviews, 17(4), 697-728.
- Loens, K., et al. (2012). Development of real-time PCR for the diagnosis of Mycoplasma pneumoniae. European Journal of Clinical Microbiology & Infectious Diseases, 31(12), 3323-3330.
- Public Health England. (2020). UK Standards for Microbiology Investigations: Detection of Mycoplasma pneumoniae.
- Centers for Disease Control and Prevention. (2021). Mycoplasma pneumoniae Infection. Retrieved from cdc.gov/pneumonia/atypical/mycoplasma.html
- Organisation for Economic Co-operation and Development. (2019). Medical Diagnostic Devices - ISO 13485:2016. Retrieved from iso.org
Related PRODUCTS
-
Precision Diagnostics for Viral Detection Using Cytomegalovirus PCR
NewsApr.16,2026 -
HPV 28 Genotyping Kit - Taizhou Cowingene Biotech Co., Ltd.|Precision & Reliability
NewsApr.14,2026 -
Advancements in PCR BK Virus Detection for Improved Patient Management
NewsApr.04,2026 -
Comprehensive Analysis of vzv PCR for Accurate Viral Detection and Diagnosis
NewsMar.31,2026 -
Comprehensive Analysis of vzv DNA PCR for Accurate Viral Detection and Diagnosis
NewsMar.28,2026 -
Comprehensive Guide to EBV DNA PCR Testing for Accurate Diagnosis and Management
NewsMar.26,2026
Sep . 08, 2025 16:39 Back to list
High-Accuracy Mycoplasma Pneumoniae Detection PCR Test
Introduction to Mycoplasma Pneumoniae Detection in B2B Diagnostics
In the landscape of clinical diagnostics and public health, the accurate and timely mycoplasma pneumoniae detection is paramount for effective patient management and disease control. Mycoplasma pneumoniae is a common cause of atypical pneumonia, particularly in children and young adults, often leading to protracted respiratory symptoms. Its subtle presentation and resistance to certain antibiotics necessitate precise diagnostic tools to guide appropriate therapeutic interventions and prevent widespread outbreaks.
For B2B stakeholders, including diagnostic laboratories, hospitals, and research institutions, investing in robust and reliable detection solutions is critical. This article delves into the technical aspects, industry trends, and practical applications of advanced diagnostic methods for Mycoplasma pneumoniae, with a focus on molecular techniques such as PCR, and introduces Cowingene's specialized offerings in this vital area.
Industry Trends in Molecular Mycoplasma Pneumoniae Detection
The diagnostic landscape for respiratory pathogens, including Mycoplasma pneumoniae, is rapidly evolving. Traditional culture-based methods, while definitive, are time-consuming and lack sensitivity for this fastidious organism. Serological tests, though quicker, often provide retrospective diagnoses due to the delay in antibody production and can suffer from cross-reactivity.
Current industry trends unequivocally point towards the widespread adoption of molecular diagnostics, particularly PCR-based assays, for their speed, sensitivity, and specificity. Key trends include:
- Automation and High-Throughput: Laboratories are seeking automated solutions that integrate sample preparation with nucleic acid amplification and detection, enabling high-volume testing with reduced hands-on time and increased efficiency.
- Multiplex PCR Panels: The ability to detect multiple respiratory pathogens simultaneously from a single sample is gaining traction, allowing for rapid differential diagnosis and optimized patient treatment, especially in distinguishing Mycoplasma pneumoniae from other viral or bacterial co-infections.
- Point-of-Care Testing (POCT): While still developing for complex molecular assays, there is a growing demand for simpler, faster, and more decentralized testing options to provide rapid results closer to the patient.
- Improved Sensitivity and Specificity: Continuous advancements in primer and probe design, as well as reaction chemistry, are leading to kits with lower limits of detection (LOD) and enhanced discriminatory power, reducing false negatives and positives. This directly impacts the reliability of mycoplasma pneumoniae detection.
- Integration with Digital Health Platforms: Seamless data transfer from diagnostic instruments to Laboratory Information Systems (LIS) and Electronic Health Records (EHR) is becoming standard, streamlining workflows and improving patient data management.
These trends underscore the critical need for advanced mycoplasma pneumoniae pcr solutions that meet the demands of modern clinical laboratories for speed, accuracy, and efficiency.

Detailed Process Flow for Mycoplasma Pneumoniae PCR Detection
The workflow for mycoplasma pneumoniae pcr test involves several critical stages, each contributing to the accuracy and reliability of the final result. For the Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid), the process emphasizes efficiency and robust performance:
-
1. Sample Collection and Preparation
Appropriate sample types for Mycoplasma pneumoniae detection include nasopharyngeal swabs, throat swabs, bronchoalveolar lavage (BAL) fluid, sputum, and sometimes urine. Samples should be collected using sterile techniques and transported in viral transport medium (VTM) or equivalent. Proper handling and storage (e.g., refrigeration at 2-8°C for short term, -70°C for long term) are crucial to preserve nucleic acid integrity. The quality of the initial sample directly impacts the success of the mycoplasma pneumoniae detection assay.
-
2. Nucleic Acid Extraction
Genomic DNA (gDNA) of Mycoplasma pneumoniae needs to be isolated from the clinical sample. This step removes inhibitors and concentrates the target nucleic acid. Automated extraction systems are commonly used in high-throughput laboratories to ensure consistency and efficiency. The Cowingene kit is compatible with various commercial nucleic acid extraction kits, adhering to international standards for nucleic acid purity and yield, often validated against ISO 13485 manufacturing processes for the extraction reagents themselves.
-
3. PCR Reaction Setup (Amplification)
The extracted DNA is then added to a reaction mixture containing the Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid). This kit contains lyophilized or liquid reagents, including highly specific primers and probes targeting conserved regions of the Mycoplasma pneumoniae genome (e.g., the P1 adhesion protein gene or 16S rRNA gene), Taq polymerase, dNTPs, and a reaction buffer. Importantly, an internal control is typically included to monitor the entire process, from extraction efficiency to PCR inhibition. This ensures the validity of negative results. The liquid format of the Cowingene kit simplifies reagent preparation and reduces pipetting errors, enhancing consistency and throughput.
-
4. Real-Time PCR Amplification and Detection
The prepared reaction mixture is loaded into a real-time PCR instrument (thermocycler). The instrument performs thermal cycling (denaturation, annealing, extension) to amplify the target DNA. During amplification, fluorescent signals from the probes are generated in real-time, which are then detected and quantified by the instrument. The software analyzes the amplification curves and cycle threshold (Ct) values to determine the presence or absence of Mycoplasma pneumoniae DNA. The presence of the internal control signal confirms the validity of the assay. Performance characteristics of the kit, such as detection limit and dynamic range, are established under rigorous quality control following ISO 13485 guidelines.
-
5. Data Analysis and Interpretation
Results are interpreted based on predefined Ct value cut-offs and the internal control performance. A positive result indicates the presence of Mycoplasma pneumoniae DNA, while a valid negative result rules out its presence within the assay's detection limits. The service life of the kit's reagents is typically specified for 12-18 months when stored correctly (e.g., -20°C). Target industries benefiting from this rapid and accurate detection method include clinical diagnostics, epidemiological surveillance, and research in respiratory infectious diseases.
The advantages in typical application scenarios include rapid diagnosis (results within hours, compared to days for culture), early intervention capabilities, and reduced spread of infection, contributing significantly to energy saving in terms of hospital resources and improved patient outcomes.
Technical Specifications: Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid)
The Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid) is engineered for high performance in molecular diagnostics, offering a sensitive and specific solution for the mycoplasma pneumoniae detection. Below are the key technical parameters and specifications:
The kit’s liquid format reduces preparation steps and potential for errors associated with lyophilized reagents, ensuring consistent performance and ease of integration into existing laboratory workflows.
Application Scenarios and Experience
The utility of accurate mycoplasma pneumoniae pcr test extends across various healthcare and research settings:
- Clinical Diagnostic Laboratories: Essential for rapid diagnosis of atypical pneumonia, guiding appropriate antibiotic therapy (e.g., macrolides instead of beta-lactams), and reducing empiric prescribing. Our kits enable labs to provide definitive results faster, improving patient outcomes.
- Hospital Infection Control: Timely detection is crucial for managing outbreaks in hospital settings, especially in pediatric wards or long-term care facilities, where Mycoplasma pneumoniae can spread rapidly. Rapid PCR helps in isolating infected individuals and preventing further transmission.
- Public Health Surveillance: Epidemiological studies and surveillance programs utilize these kits to monitor disease prevalence, identify emerging strains, and track resistance patterns, contributing to public health strategies.
- Academic and Research Institutions: Researchers employ these kits for studies on pathogen virulence, host immune responses, and the development of new diagnostic or therapeutic interventions.
Customer Feedback (Simulated): "Our laboratory implemented the Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid) six months ago, and the transition was seamless. The liquid format significantly reduced our prep time, and the consistency of results has been outstanding. We've seen a noticeable improvement in our turnaround times for mycoplasma pneumoniae detection, which directly benefits our clinicians and patients." – Dr. L. Chen, Head of Molecular Diagnostics, Regional Hospital Group.

Technical Advantages of PCR-Based Mycoplasma Pneumoniae Detection
Compared to conventional methods, PCR-based mycoplasma pneumoniae detection offers significant technical advantages:
- High Sensitivity: PCR can detect very low concentrations of bacterial DNA, often before the host mounts a significant immune response or before the organism can be cultured, enabling early diagnosis. This is crucial for preventing disease progression and spread.
- Exceptional Specificity: Designed primers and probes ensure that only Mycoplasma pneumoniae DNA is amplified, minimizing false positives that can occur due to cross-reactivity with other common respiratory flora or pathogens, which is a common issue with serological methods.
- Rapid Turnaround Time: While culture can take weeks, and serology relies on antibody kinetics, a mycoplasma pneumoniae pcr test can provide results within hours of sample reception, facilitating timely clinical decisions.
- Broad Detection Window: PCR can detect the pathogen during both acute and convalescent phases of infection, making it useful throughout the disease course, unlike serology which is often limited to later stages.
- Reliable Internal Control: The inclusion of an internal control in kits like Cowingene’s ensures that negative results are truly negative and not due to extraction failures or PCR inhibition, providing robust quality assurance.
- Enhanced Workflow Efficiency: Modern PCR kits, especially those in liquid format, are designed for ease of use, compatibility with automated systems, and reduced hands-on time, optimizing laboratory throughput.
Cowingene's kit specifically offers the advantage of a liquid format, which improves pipetting accuracy, reduces preparation steps, and minimizes reagent waste, contributing to a more streamlined and cost-effective workflow for laboratories.
Vendor Comparison: Mycoplasma Pneumoniae PCR Kits
Choosing the right mycoplasma pneumoniae pcr test kit requires careful consideration of various factors, including performance, ease of use, regulatory compliance, and cost-effectiveness. Below is a comparative overview of typical parameters across different vendors, highlighting Cowingene's competitive positioning:
Cowingene's kit stands out with its optimal balance of sensitivity, specificity, and user-friendly liquid format, making it a highly competitive choice for laboratories prioritizing efficiency and reliable results.
Customized Solutions and Integration
Recognizing that every laboratory has unique operational requirements, Cowingene offers flexible solutions and integration support for its mycoplasma pneumoniae detection kits. Our team of technical specialists can assist in:
- Workflow Optimization: Advising on the seamless integration of our kits with existing nucleic acid extraction platforms (manual or automated) and real-time PCR instruments.
- LIMS/LIS Integration: Providing support for data output compatibility to ensure smooth integration with Laboratory Information Management Systems (LIMS) and Laboratory Information Systems (LIS) for efficient data management and reporting.
- Training and Technical Support: Offering comprehensive training for laboratory personnel on kit usage, troubleshooting, and best practices to maximize assay performance and ensure robust mycoplasma pneumoniae pcr testing.
- Bulk and Customized Packaging: For high-volume users or specific research needs, we can discuss customized packaging or bulk reagent supply to meet specific demands, all while maintaining strict quality controls aligned with ISO standards.
- Regional Adaptations: Collaborating with local partners to ensure kits meet specific regional regulatory requirements or endemic pathogen profiles, enhancing local relevance and adoption.
Our commitment to partnership ensures that laboratories can leverage our diagnostic solutions effectively, irrespective of their scale or operational complexities.

Application Case Studies
Case Study 1: Rapid Diagnosis in a Pediatric Hospital
A prominent children's hospital frequently encountered cases of atypical pneumonia. Prior to implementing advanced molecular diagnostics, presumptive treatment was common, leading to suboptimal antibiotic use and prolonged hospitalization for some patients. After integrating Cowingene's Mycoplasma Pneumoniae Detection Kit (Liquid) into their routine diagnostics, the hospital observed a significant improvement.
- Challenge: Slow turnaround times for Mycoplasma pneumoniae diagnosis (3-5 days for serology, weeks for culture).
- Solution: Implementation of Cowingene's mycoplasma pneumoniae pcr test, enabling same-day results post-sample collection.
- Outcome: Diagnosis time was reduced by over 80%. This led to a 30% reduction in inappropriate broad-spectrum antibiotic prescriptions for Mycoplasma pneumoniae cases, a 25% decrease in average hospital stay for affected children, and improved infection control measures due to rapid identification. The high sensitivity of the kit prevented false negatives, ensuring accurate clinical decisions.
Case Study 2: Outbreak Investigation in a University Dormitory
During a respiratory illness outbreak in a university dormitory, public health authorities needed to quickly identify the causative agent to prevent further spread. Initial viral panels were negative, prompting a focus on atypical bacterial pathogens.
- Challenge: Rapid identification of the pathogen during an active outbreak with a high potential for rapid transmission among close contacts.
- Solution: Rapid deployment of Cowingene's mycoplasma pneumoniae detection kit on collected nasopharyngeal swabs from symptomatic individuals. The kit's liquid format and ease of use allowed for quick scaling of testing.
- Outcome: Within 24 hours of sample collection, Mycoplasma pneumoniae was confirmed in 70% of symptomatic students. This rapid, authoritative identification allowed public health officials to swiftly implement targeted interventions, including contact tracing, prophylactic treatment for close contacts, and specific health advisories, effectively containing the outbreak and minimizing disruption to university operations. The accuracy prevented misdiagnosis and unnecessary broad-spectrum interventions.
Ensuring Trust: Certifications and Quality Assurance
Cowingene is committed to providing high-quality, reliable diagnostic solutions. Our Mycoplasma Pneumoniae Detection Kit (Liquid) undergoes rigorous quality control and adheres to international standards:
- CE-IVD Marking: The kit is CE-IVD marked, indicating conformity with the European In Vitro Diagnostic Medical Devices Regulation (IVDR), ensuring it meets stringent safety and performance requirements for use in the European Union.
- ISO 13485 Certification: Our manufacturing facilities and quality management system are certified to ISO 13485:2016 standards, demonstrating our commitment to producing medical devices safely and effectively. This ensures every batch of mycoplasma pneumoniae pcr kit maintains consistent quality.
- Comprehensive Validation Data: Each kit lot is extensively tested for sensitivity, specificity, accuracy, and reproducibility against reference panels and clinical samples. Detailed validation reports and performance data are available upon request.
- Traceability: All raw materials and manufacturing processes are fully traceable, ensuring transparency and accountability at every stage of production.
These certifications and internal quality protocols underline our dedication to providing authoritative and trustworthy tools for mycoplasma pneumoniae detection, supporting clinical decision-making with confidence.
Trustworthiness: FAQ, Lead Time, Warranty, and Support
Frequently Asked Questions (FAQ)
-
Q: What is the shelf life of the Cowingene Mycoplasma Pneumoniae Detection Kit (Liquid)?
A: The kit has a shelf life of 12 months from the date of manufacture when stored at -20°C in the dark.
-
Q: Is the kit compatible with automated nucleic acid extraction systems?
A: Yes, our kit is validated for compatibility with several automated extraction systems. Please contact our technical support for a list of validated systems.
-
Q: How does the internal control function in the mycoplasma pneumoniae pcr test?
A: The internal control detects potential PCR inhibition and confirms successful nucleic acid extraction, ensuring the reliability of negative results.
-
Q: Can this kit differentiate between Mycoplasma pneumoniae and other Mycoplasma species?
A: Yes, the primers and probes are highly specific to Mycoplasma pneumoniae and do not cross-react with other common Mycoplasma species or respiratory pathogens, ensuring accurate mycoplasma pneumoniae detection.
Lead Time and Fulfillment
Cowingene maintains a robust supply chain to ensure timely delivery of our diagnostic kits. Standard lead time for most orders is 5-7 business days within major regions. For large volume orders or custom solutions, lead times will be communicated upon order confirmation. We work with established logistics partners to ensure product integrity during transit.
Warranty Commitments
Cowingene guarantees that its Mycoplasma Pneumoniae Detection Kit (Liquid) will perform according to the specifications provided in the product insert for the duration of its stated shelf life, when stored and used as directed. Any product found to be defective under these conditions will be replaced or refunded. Full warranty terms are detailed in our product documentation.
Customer Support
Our dedicated customer support and technical service teams are available to assist with product inquiries, assay optimization, troubleshooting, and training. We offer support via phone, email, and online resources. Our goal is to ensure your laboratory experiences seamless integration and optimal performance with our mycoplasma pneumoniae pcr test.
Contact Information:
- Email: sales@cowingene.com
- Phone: +86-571-8888-8888 (Simulated)
- Website: www.cowingene.com

Conclusion
The demand for accurate, rapid, and reliable mycoplasma pneumoniae detection is continually growing, driven by evolving clinical needs and the desire for improved patient outcomes. Molecular diagnostic tools, particularly real-time PCR, represent the gold standard for this challenge. Cowingene's Mycoplasma Pneumoniae Detection Kit (Liquid) offers a robust, highly sensitive, and specific solution, designed with the B2B laboratory's efficiency and reliability in mind. By adhering to stringent quality standards, providing comprehensive support, and embracing innovative formats, Cowingene empowers healthcare providers and researchers with the tools necessary for effective management of Mycoplasma pneumoniae infections.
References
- Waites, K. B., & Talkington, D. F. (2004). Mycoplasma pneumoniae and its role as a human pathogen. Clinical Microbiology Reviews, 17(4), 697-728.
- Loens, K., et al. (2012). Development of real-time PCR for the diagnosis of Mycoplasma pneumoniae. European Journal of Clinical Microbiology & Infectious Diseases, 31(12), 3323-3330.
- Public Health England. (2020). UK Standards for Microbiology Investigations: Detection of Mycoplasma pneumoniae.
- Centers for Disease Control and Prevention. (2021). Mycoplasma pneumoniae Infection. Retrieved from cdc.gov/pneumonia/atypical/mycoplasma.html
- Organisation for Economic Co-operation and Development. (2019). Medical Diagnostic Devices - ISO 13485:2016. Retrieved from iso.org
Related PRODUCTS








