Oct . 24, 2025 09:46 Back to list
Respiratory Panel Test for Fast, Accurate Pathogen Detection
SARS‑CoV‑2/Flu A/B multiplex kits are quietly reshaping respiratory diagnostics
If you’ve been shopping for a respiratory panel test for rapid, syndromic triage in busy seasons, you’ve probably noticed one theme: lyophilized (freeze‑dried) chemistry is winning. To be honest, it’s not just a fad—labs like the stability and the no‑ice‑pack convenience. Cowingene’s SARS‑CoV‑2 & Influenza A/B Detection Kit (Lyophilized) rides that trend with a pragmatic spec sheet and an old‑school focus on compatibility over hype.
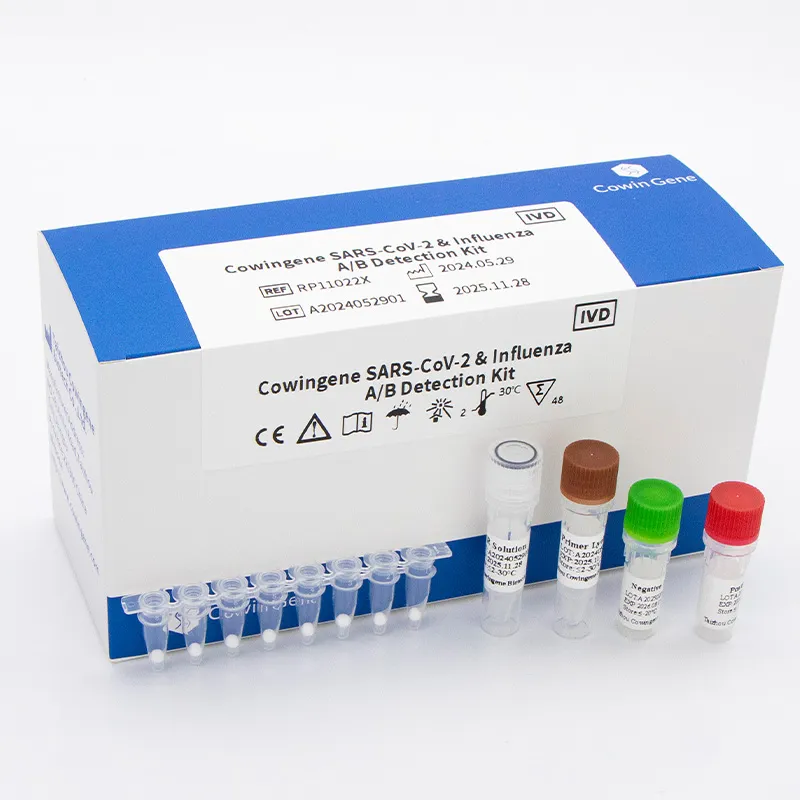
What it is, who it’s for
Product: Cowingene SARS‑CoV‑2 & Influenza A/B Detection Kit (Lyophilized), REF: RP11022X. Validated specimens: nasopharyngeal swab/aspirate, bronchoalveolar lavage, throat swab. Targets: SARS‑CoV‑2, Flu A, Flu B by RT‑qPCR. In short, a compact three‑plex for frontline respiratory season. Many customers say the lyophilized format feels less fussy—no cold‑chain panic—especially in satellite clinics.
Product specifications (key points)
| Format | Lyophilized RT‑qPCR master mix with target‑specific primers/probes |
| Analytes | SARS‑CoV‑2, Influenza A, Influenza B |
| Validated specimens | Nasopharyngeal swab/aspirate, BAL, throat swab |
| Instruments | Common qPCR platforms with FAM/HEX/ROX (or equivalent) channels; check IFU for channel mapping |
| Storage & shelf life | Lyophilized stability helps; typical shelf life ≈ 12–18 months (real‑world use may vary; follow label) |
| Origin | NO.28, Xinlin Road, Taizhou city, Jiangsu Province, China |
| REF | RP11022X |
Industry trends you can’t ignore
- Multiplexing: one swab, several answers—cuts turnaround and conserves reagents.
- Lyophilization: better logistics, fewer cold‑chain headaches, steadier field performance.
- Standards‑first validation: labs cite CLSI EP17 for LoD and ISO 15189 workflows as table stakes now.
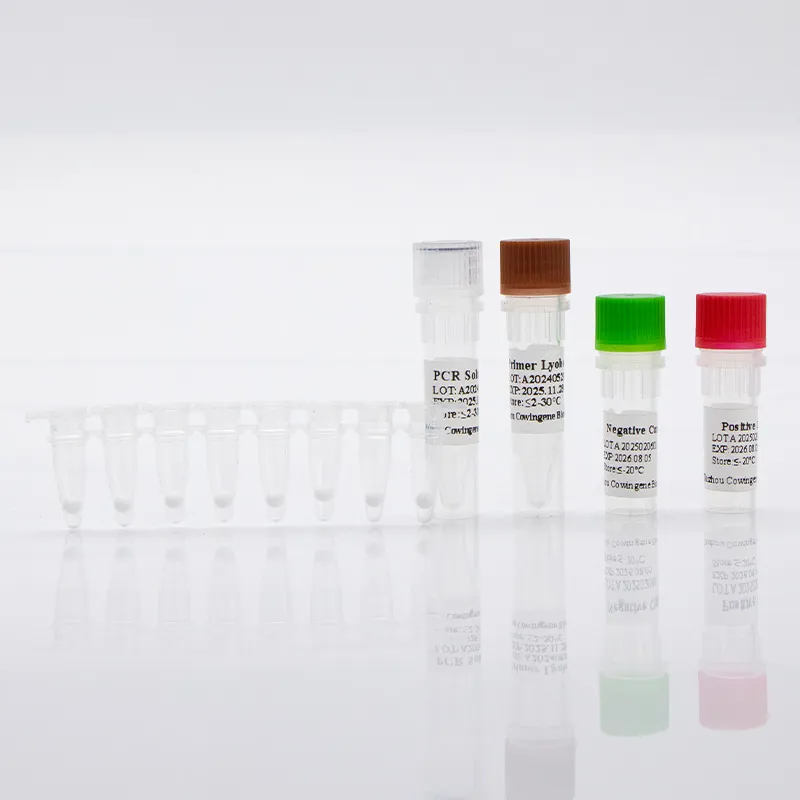
Where it fits (use cases)
ER triage during peak flu/COVID waves; employer/school outbreaks; small private labs lacking ultra‑cold storage; mobile screening where power is patchy. A lab lead told me—off the record—that lyophilized beads “just travel better.” I guess that tracks.
Process flow (materials, methods, standards)
- Materials: validated swabs/transport media; extraction kit (or validated extraction‑free workflow); lyophilized tube/plate; nuclease‑free water.
- Method: rehydrate lyophilized master mix; add extracted RNA; RT‑qPCR with proper channel settings; include negative/positive controls.
- Standards: verify LoD per CLSI EP17; precision per CLSI EP05; qualitative verification per CLSI MM17; lab QMS under ISO 15189; manufacturing under ISO 13485.
- Service life: follow lot label; avoid humidity spikes; rehydrated mixes typically single‑use.
- Industries: hospitals, public health labs, aviation ports, campus health, contract testing.
Vendor snapshot (approximate, check IFUs)
| Vendor/Kit | Format | Targets | Storage | Pros | Considerations |
|---|---|---|---|---|---|
| Cowingene SARS‑CoV‑2/Flu A/B (Lyophilized) | Lyophilized RT‑qPCR | SARS‑CoV‑2, Flu A, Flu B | Ambient‑tolerant (≈) | Cold‑chain light; flexible instruments | Rehydration timing matters |
| Thermo Fisher TaqPath COVID‑19/Flu A/B (example) | Liquid RT‑qPCR | SARS‑CoV‑2, Flu A, Flu B | Cold chain | Broad ecosystem | More freezer space |
| Roche cobas SARS‑CoV‑2 & Flu A/B (example) | System‑integrated | SARS‑CoV‑2, Flu A, Flu B | System‑defined | Automation, throughput | Platform lock‑in |
Note: compiled from public factsheets as of publication; specifications and availability may change; real‑world performance varies by lab protocol.
Customization and feedback
- OEM/ODM: private labeling, kit size adjustments, alternative dye sets—often feasible on request.
- Workflow tweaks: some labs validate extraction‑free on high‑viral‑load swabs; others tighten Ct thresholds (e.g., ≤38) for specificity.
- Customer notes: users like the consistent bead rehydration; a few suggest letting tubes sit 3–5 minutes before mixing for smoother Ct curves.
Performance context
In literature, well‑designed RT‑qPCR respiratory panels report LoD bands around 200–1000 copies/mL and repeatability with Ct drift typically
If you’re shortlisting a respiratory panel test for winter surge planning, another respiratory panel test for decentralized sites, or even a respiratory panel test for travel‑adjacent screening, this kit’s lyophilized angle is, frankly, practical.
Compliance checkpoints
- Manufacturer QMS: ISO 13485.
- Clinical lab competence: ISO 15189.
- Method verification: CLSI EP17 (LoD), EP05 (precision), MM17 (molecular assay verification).
- Market access: check local regulations (e.g., EU IVDR, national authorities).
Citations
- World Health Organization. Diagnostic testing for SARS‑CoV‑2. https://www.who.int/publications
- CLSI EP17-A2. Evaluation of Detection Capability for Clinical Laboratory Measurement Procedures. https://clsi.org
- ISO 13485:2016 Medical devices—Quality management systems. https://www.iso.org
- ISO 15189:2022 Medical laboratories—Requirements for quality and competence. https://www.iso.org
- Regulation (EU) 2017/746 (IVDR). https://eur-lex.europa.eu
Related PRODUCTS
-
Rapid HPV 16/18/45 Molecular Detection with NATBox for Targeted Cervical Cancer Screening
NewsApr.24,2026 -
High-Sensitivity HPV (2+16) Molecular Detection for Early Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 28 Genotyping Solution for Advanced Cervical Cancer Screening
NewsApr.24,2026 -
Comprehensive HPV 15 Genotyping Solution for Accurate Cervical Cancer Risk Assessment
NewsApr.24,2026 -
Comprehensive 14-Pathogen STI Detection Solution for Accurate Molecular Diagnosis
NewsApr.24,2026 -
Accurate and Reliable HSV-1/2 Molecular Detection for Clinical Diagnosis
NewsApr.24,2026








