apr. . 24, 2026 13:53 Înapoi la listă
Accurate and Reliable HSV-1/2 Molecular Detection for Clinical Diagnosis
Herpes Simplex Virus (HSV) infection is one of the most prevalent viral infections worldwide and remains a significant concern in the field of sexually transmitted infections (STIs). Due to its lifelong persistence, potential for recurrence, and often asymptomatic presentation, HSV poses a continuous challenge for clinical diagnosis and infection control. Without timely detection and proper management, HSV infections may lead to complications such as neonatal transmission, increased susceptibility to other infections, and serious reproductive health issues.
The Kit de detectare a virusului Herpes Simplex Cowingene 1/2 (liofilizat) is an advanced in vitro diagnostic (IVD) solution designed for the qualitative detection of HSV-1 and HSV-2. Based on real-time PCR technology, this kit enables rapid, sensitive, and specific identification of viral DNA, providing reliable support for clinical diagnosis and treatment of genitourinary tract infections.
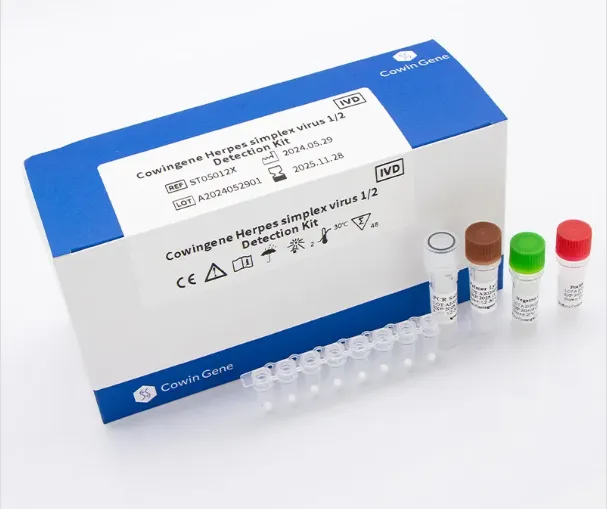
Intended Use: Targeted HSV Detection for Clinical Decision Support
This detection kit is specifically intended for the qualitative detection of Herpes Simplex Virus type 1 (HSV-1) and type 2 (HSV-2) in human clinical samples. It provides important auxiliary information for clinicians in diagnosing HSV-related infections and guiding therapeutic strategies.
HSV is primarily transmitted through close contact:
HSV-1 is commonly spread via oral contact and causes oral herpes (cold sores), but can also lead to genital infections
HSV-2 is mainly transmitted through sexual contact and is the leading cause of genital herpes
Given the overlapping clinical symptoms and differences in transmission patterns, accurate differentiation between HSV-1 and HSV-2 is critical for effective patient management and epidemiological control.
Flexible Sample Types: Adapted to Real Clinical Needs
The kit is validated for use with multiple clinically relevant specimen types:
Urine samples
Male urethral swabs
Female cervical swabs
Female vaginal swabs
This multi-sample compatibility allows healthcare providers to choose appropriate collection methods based on patient conditions, improving diagnostic flexibility and patient compliance. It is suitable for both clinician-collected and self-collected samples, supporting broader screening applications.
Performance Characteristics: High Sensitivity and Diagnostic Accuracy
High Sensitivity for Early Detection
The kit achieves a limit of detection (LoD) of 400 copies/mL, ensuring reliable detection of HSV DNA even at low viral loads. This is particularly important for early-stage infections and asymptomatic carriers, where viral concentration may be minimal.
Accurate Differentiation of HSV-1 and HSV-2
Using multiplex real-time PCR technology, the kit enables simultaneous detection and differentiation of HSV-1 and HSV-2 within a single reaction tube. This improves diagnostic efficiency and reduces testing time.
Reliable and Reproducible Results
The PCR-based detection system ensures high specificity and minimizes false-positive or false-negative results. It provides stable and consistent performance across different laboratory conditions.
Advanced Technical Specifications for Modern Laboratories
Detection Method: Real-time PCR (single-tube multiplex detection)
Detection Targets: HSV-1 and HSV-2
Detection Channels: FAM, VIC/HEX, ROX, CY5
Compatible Instruments:
ABI 7500
Roche LightCycler 480
Bio-Rad CFX96
Other open real-time PCR systems
Storage Conditions: 2–30°C
Shelf Life: 18 months
Certification: CE-IVD
These specifications ensure compatibility with mainstream PCR platforms and facilitate easy integration into existing laboratory workflows.
Lyophilized Reagent Advantage: Stability and Convenience
One of the key highlights of this kit is its lyophilized (freeze-dried) format, which offers significant operational and logistical advantages:
Room temperature storage (2–30°C) reduces dependence on cold-chain transportation
Extended shelf life (18 months) ensures long-term usability
Simplified workflow with minimal preparation steps
Reduced risk of contamination and handling errors
This makes the kit especially suitable for decentralized laboratories, resource-limited settings, and point-of-care testing environments.
Streamlined Workflow with Supporting Reagents
The kit can be integrated into a complete molecular diagnostic workflow using compatible supporting reagents, including:
Viral DNA/RNA extraction kits
Sample release reagents
Self-collection kits for urine and vaginal samples
This ensures a standardized and efficient testing process, from sample collection to nucleic acid extraction and amplification, improving laboratory throughput and consistency.
Key Advantages for Clinical and Laboratory Applications
Targeted HSV detection with clear differentiation between HSV-1 and HSV-2
High sensitivity (LoD 400 copies/mL) for early infection detection
Multiple validated sample types for flexible clinical use
Lyophilized format for enhanced stability and easy transport
Wide instrument compatibility for seamless lab integration
Simplified workflow to improve efficiency and reduce errors
CE-IVD certified for quality assurance and regulatory compliance
Conclusion: A Reliable Molecular Tool for Herpes Diagnosis
The Cowingene Herpes Simplex Virus 1/2 Detection Kit (Lyophilized) provides a highly reliable, accurate, and user-friendly molecular diagnostic solution for HSV detection. By combining real-time PCR technology with lyophilized reagent stability, it addresses key challenges in herpes diagnosis, including sensitivity, workflow efficiency, and logistical limitations.
This kit supports early detection, accurate differentiation, and timely clinical intervention, ultimately contributing to improved patient outcomes and more effective management of sexually transmitted infections.
As healthcare systems continue to emphasize rapid and precise diagnostics, this HSV-1/2 detection kit stands out as a valuable tool for modern laboratories and clinical settings worldwide.
Înrudit PRODUSE
-
Rapid HPV 16/18/45 Molecular Detection with NATBox for Targeted Cervical Cancer Screening
ŞtiriApr.24,2026 -
High-Sensitivity HPV (2+16) Molecular Detection for Early Cervical Cancer Screening
ŞtiriApr.24,2026 -
Comprehensive HPV 28 Genotyping Solution for Advanced Cervical Cancer Screening
ŞtiriApr.24,2026 -
Comprehensive HPV 15 Genotyping Solution for Accurate Cervical Cancer Risk Assessment
ŞtiriApr.24,2026 -
Comprehensive 14-Pathogen STI Detection Solution for Accurate Molecular Diagnosis
ŞtiriApr.24,2026








