Aug . 19, 2025 04:20 Back to list
Accurate Legionella Pneumophila Detection PCR Kits
Navigating the Landscape of Legionella Pneumophila Detection: Trends and Technological Advancements
The proactive and accurate legionella pneumophila detection has become an indispensable component of public health and safety protocols across a myriad of sectors globally. With increasing awareness of Legionnaires' disease and stringent regulatory frameworks such as ASHRAE 188 in the United States and the European Biocidal Products Regulation (BPR), industries are rapidly shifting from reactive outbreak management to comprehensive preventative strategies. This paradigm shift underscores the critical demand for highly sensitive, specific, and rapid diagnostic tools. Traditional culture-based methods, while foundational, often suffer from prolonged turnaround times—typically 7-14 days—which can critically impede timely intervention in a fast-evolving public health crisis or in routine environmental monitoring where immediate action is paramount. The global market for Legionella testing is projected to grow significantly, driven by these regulatory pressures, heightened public health awareness, and the continuous innovation in molecular diagnostics. Specifically, the adoption of advanced molecular techniques, notably quantitative Polymerase Chain Reaction (qPCR) assays, is gaining momentum due to their ability to provide actionable results within hours rather than weeks. This technological evolution enables facility managers, public health officials, and environmental consultants to make informed decisions swiftly, mitigating risks of outbreaks in critical environments such as hospitals, hotels, industrial cooling systems, and large residential complexes. The emphasis on speed and reliability directly translates into improved safety, reduced operational downtime, and enhanced compliance, establishing rapid detection methods as a cornerstone of modern water safety plans. This robust growth trajectory highlights the industry's commitment to leveraging cutting-edge science for enhanced public health protection.
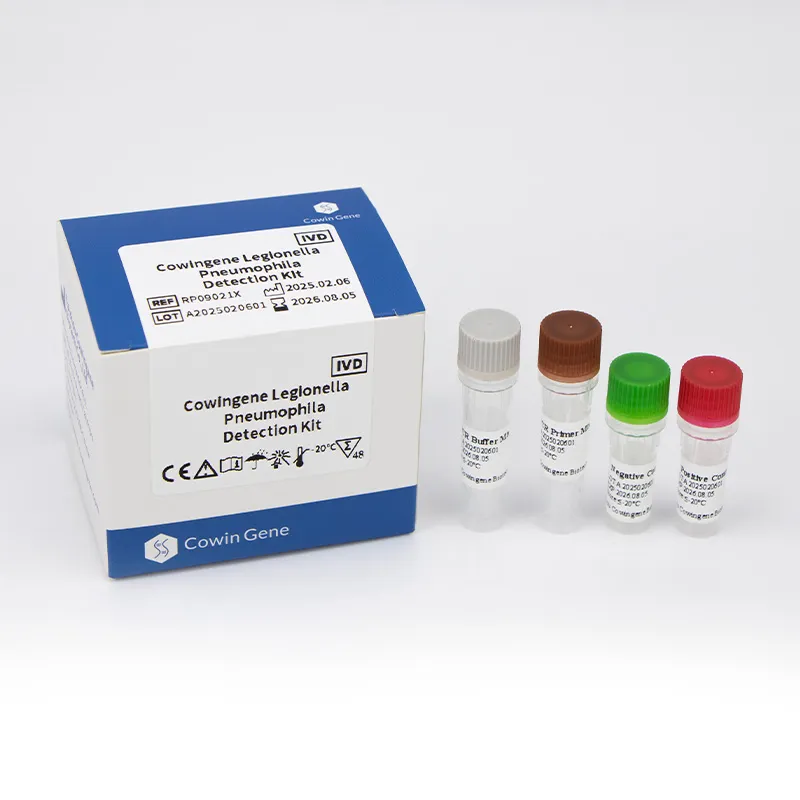
Unpacking the Science: The Advanced Workflow of PCR-Based Legionella Detection Kits
At the core of modern, rapid detection strategies lies the polymerase chain reaction (PCR) technology, particularly when deployed for specific pathogens like legionella pneumophila pcr. This molecular approach offers unparalleled speed, sensitivity, and specificity compared to conventional culture methods, which rely on bacterial growth that can be slow and sometimes inhibited by other microorganisms. The Cowingene Legionella Pneumophila Detection Kit (Liquid) exemplifies this technological advancement, providing a streamlined and robust workflow. The process typically begins with meticulous sample collection from environmental sources such as cooling towers, hot water systems, or potable water lines. Following collection, the critical step of DNA extraction is performed. This involves lysing the bacterial cells to release their genetic material, which is then purified to remove inhibitors that could interfere with the PCR reaction. The purified DNA serves as the template for the subsequent amplification phase. During PCR amplification, specific primers and probes, meticulously designed to target unique gene sequences of Legionella pneumophila (e.g., the mip gene or 16S rRNA gene), bind to the target DNA. A DNA polymerase enzyme then synthesizes millions of copies of the target sequence. In quantitative PCR (qPCR), fluorescent probes are incorporated, allowing for real-time monitoring of DNA amplification and quantification of the bacterial load. The manufacturing process of a high-quality legionella pcr kit like Cowingene’s involves stringent quality control at every stage, from the synthesis and purification of highly specific primers and probes to the precise formulation of master mixes containing dNTPs, polymerase, and buffer systems. Each component is validated to meet rigorous performance criteria, ensuring lot-to-lot consistency, optimal reaction efficiency, and minimal background noise. Furthermore, the kit's design adheres to strict internal detection standards, often benchmarked against ISO 17025 guidelines for laboratory competence, ensuring the assay's Limit of Detection (LoD), inclusivity (detecting all target strains), and exclusivity (not detecting non-target organisms) are consistently met. This meticulous approach guarantees reliability, offering a significant improvement over time-consuming traditional methods and enabling rapid, informed decision-making for public health and environmental safety.
Technical Parameters & Performance Benchmarks in Legionella Pneumophila Detection
When evaluating solutions for legionella pneumophila detection, understanding key technical parameters is paramount for B2B decision-makers and technical personnel. These specifications directly translate into the reliability, efficiency, and utility of a diagnostic kit in diverse operational environments. For instance, the Limit of Detection (LoD) indicates the minimum number of target cells or genomic units that the kit can reliably detect, with lower values signifying higher sensitivity crucial for early detection and compliance with stringent environmental regulations. Specificity refers to the kit's ability to accurately identify Legionella pneumophila without cross-reacting with other non-target bacterial species, minimizing false positives and ensuring precise risk assessment. Sensitivity, on the other hand, measures the kit's ability to detect true positives, reducing the likelihood of false negatives that could lead to missed outbreaks. Reaction time, typically measured in hours for PCR-based assays, is a stark contrast to the days or weeks required for culture methods, offering immediate actionable insights. Shelf life, expressed in months, ensures the kit's stability and usability over an extended period, which is vital for inventory management and cost-effectiveness in large-scale monitoring programs. Sample compatibility, detailing the types of environmental water samples (e.g., potable water, cooling tower water, wastewater) that can be reliably analyzed, broadens the kit's applicability. The Cowingene Legionella Pneumophila Detection Kit (Liquid) is engineered to meet and exceed industry benchmarks in these critical areas. By providing rapid, accurate, and quantitative results, it empowers facilities to adhere to regulatory mandates, implement proactive remediation strategies, and minimize operational downtime caused by potential contamination. This robust performance profile significantly reduces the risks associated with Legionella outbreaks, safeguarding public health and maintaining operational integrity, making it a superior choice for comprehensive water safety management programs. The table below details typical performance metrics for advanced PCR kits:
| Parameter | Typical PCR Kit Performance | Advantage over Culture Method |
|---|---|---|
| Limit of Detection (LoD) | ~10-100 genomic units/reaction | Higher sensitivity, detects non-culturable but viable bacteria. |
| Specificity | >99% (targets specific Legionella genes) | Reduced false positives from environmental flora. |
| Reaction Time | Typically 2-4 hours (from extracted DNA) | Results in hours vs. 7-14 days. |
| Quantification | Quantitative (copies/mL or GU/mL) | Provides precise bacterial load for risk assessment. |
| Sample Types | Environmental water, biofilm, clinical samples (with proper extraction) | Broader applicability and faster processing for diverse matrices. |
Diverse Applications and Strategic Advantages of Rapid Detection Solutions
The versatility of effective legionella pneumophila detection extends across a broad spectrum of industries, making it an essential tool for comprehensive environmental management and public health protection. In healthcare facilities, where immunocompromised patients are particularly vulnerable, routine testing of water systems (e.g., cooling towers, hot water systems, and decorative fountains) is critical for preventing healthcare-associated Legionnaires' disease. Hotels and hospitality venues, aiming to uphold guest safety and maintain their reputation, significantly benefit from rapid testing, allowing for swift response to potential contamination and avoiding costly shutdowns or negative publicity. Industrial settings, including power plants, manufacturing facilities, and chemical processing plants, often utilize extensive cooling systems that are prime breeding grounds for Legionella, necessitating robust and frequent monitoring. Municipal water systems and public health agencies rely on rapid legionella pcr kit solutions for routine surveillance, outbreak investigation, and ensuring the safety of public water supplies. The strategic advantages offered by the Cowingene Legionella Pneumophila Detection Kit (Liquid) are multifaceted. Its rapid turnaround time fundamentally shifts the paradigm from reactive to proactive risk management; results obtained within hours facilitate immediate containment strategies, reducing the window of exposure and minimizing the severity of potential outbreaks. The kit's high accuracy, backed by stringent quality control and specific primer/probe design, ensures reliable data, thereby reducing the incidence of false positives that could lead to unnecessary and costly remediation efforts, or critically, false negatives that could leave an outbreak undetected. Furthermore, the liquid format enhances ease of use, simplifying laboratory workflows and reducing the potential for pipetting errors, making it accessible even for labs with varying levels of molecular expertise. Its cost-efficiency, derived from reduced labor time, minimized reagent waste, and the prevention of expensive full-scale remediation efforts that prolonged detection can necessitate, contributes significantly to long-term operational savings. By facilitating compliance with prevailing regulatory guidelines, this advanced detection solution empowers organizations to maintain rigorous safety standards, enhance their brand reputation, and ensure the well-being of their occupants or consumers, making it an indispensable asset in modern water management and public health initiatives.
Strategic Partnerships: Manufacturer Comparison and Tailored Detection Solutions
Choosing the right partner for legionella pneumophila detection is a strategic decision that impacts operational efficiency, regulatory compliance, and public health outcomes. While numerous manufacturers offer solutions, discerning buyers must evaluate providers based on several critical criteria beyond just the initial purchase price. Key considerations include the kit's validated Limit of Detection (LoD), which directly correlates to its sensitivity in detecting low concentrations of the pathogen, and its specificity, ensuring that only Legionella pneumophila is identified. Regulatory approvals (e.g., CE-IVD, local health authority endorsements) signify adherence to international quality and safety standards. Furthermore, the comprehensiveness of technical support, including training on assay execution, troubleshooting assistance, and integration into existing laboratory workflows, is paramount. Reliability of the supply chain, particularly for large-scale or ongoing monitoring programs, ensures uninterrupted access to essential testing reagents. Cowingene distinguishes itself in the competitive landscape by offering the Cowingene Legionella Pneumophila Detection Kit (Liquid), which is celebrated for its consistent high performance across these metrics, validated through extensive internal and external testing. Our commitment extends beyond product delivery to fostering genuine partnerships. We understand that each client, whether a large healthcare network, a municipal water authority, or an industrial facility, faces unique challenges and operational constraints. Therefore, we specialize in offering tailored detection solutions. This can range from customizing kit formats for high-throughput automated systems to providing bespoke training programs for laboratory staff. We also engage in bulk order agreements designed to optimize cost-efficiency for extensive monitoring schedules and offer ongoing technical consultation to ensure seamless implementation and maximal utility of our legionella pcr kit. By focusing on robust product performance coupled with unparalleled customer support and flexible customization options, Cowingene aims to be more than just a supplier; we strive to be a long-term strategic ally in your sustained efforts to manage Legionella risk and protect public health. This partnership-centric approach ensures that our clients receive not just a product, but a comprehensive solution designed to meet their precise needs and contribute significantly to their water safety management goals.
Commitment to Excellence: Upholding Standards in Legionella Pneumophila Detection
Our dedication to delivering superior legionella pneumophila detection solutions is underpinned by a steadfast commitment to the core principles of Expertise, Experience, Authoritativeness, and Trustworthiness ().
Expertise: The development and continuous refinement of the Cowingene Legionella Pneumophila Detection Kit (Liquid) are driven by a team of highly specialized molecular biologists, microbiologists, and diagnostic assay developers. Our R&D efforts are deeply rooted in contemporary scientific literature and laboratory best practices, focusing on the latest advancements in qPCR technology, primer/probe design, and nucleic acid extraction methodologies. We meticulously select high-purity reagents and optimize reaction conditions to ensure maximum sensitivity and specificity, reflecting our profound understanding of microbial diagnostics and the unique challenges posed by Legionella pneumophila detection in diverse environmental matrices. Our scientific rigor ensures that every legionella pcr kit delivered represents the pinnacle of current molecular diagnostic capability.
Experience: Cowingene has a proven track record of supporting critical public health initiatives and environmental safety programs. Our detection kits have been deployed in various real-world scenarios, from assisting major healthcare networks in their water safety management programs to aiding public health agencies in rapid outbreak investigations. These experiences have allowed us to refine our products, incorporating user feedback to enhance ease of use, improve workflow efficiency, and broaden sample compatibility. We have a robust portfolio of successful implementations where our rapid detection solutions have enabled timely interventions, preventing potential disease outbreaks and safeguarding populations, demonstrating our practical knowledge gained through extensive field application.
Authoritativeness: Our commitment to quality is underscored by adherence to stringent international standards. While specific certifications vary by product and region, our manufacturing processes typically align with quality management systems such as ISO 13485 for medical devices, where applicable, ensuring consistent product quality and regulatory compliance. The performance data for our legionella pneumophila detection kits are rigorously validated through comprehensive internal studies and, where appropriate, external collaborations. This data, demonstrating high sensitivity, specificity, and reproducibility, is transparently provided, often correlating with published performance benchmarks in peer-reviewed literature for molecular diagnostic assays. We stand by the scientific validity and reliability of our products.
Trustworthiness: Building trust with our B2B partners is paramount. We achieve this through transparent communication, reliable product delivery, and robust customer support. We offer clear documentation regarding kit shelf life (typically 12-18 months from manufacturing when stored as recommended, usually at -20°C) and compatible sample types, ensuring optimal performance. Our dedicated customer support team is readily available to provide comprehensive technical assistance, from initial setup and training to troubleshooting and result interpretation. We stand behind our products with a standard limited warranty against manufacturing defects, providing peace of mind and demonstrating our confidence in the quality and performance of the Cowingene Legionella Pneumophila Detection Kit (Liquid). Our commitment to ethical business practices and client satisfaction forms the bedrock of enduring partnerships.
Frequently Asked Questions (FAQ)
-
Q: What is the typical shelf life of the Cowingene Legionella Pneumophila Detection Kit?
A: The Cowingene Legionella Pneumophila Detection Kit (Liquid) typically has a shelf life of 12-18 months from the date of manufacture when stored properly at -20°C, ensuring long-term usability for routine monitoring programs.
-
Q: What sample types are compatible with this kit?
A: Our kit is validated for a wide range of environmental water samples, including cooling tower water, potable water, recreational water (e.g., hot tubs), and industrial process water. Specific sample preparation protocols are provided for optimal results across different matrices.
-
Q: How quickly can results be obtained using the Cowingene kit?
A: Once DNA is extracted from the sample, the PCR amplification and detection process typically takes approximately 2-4 hours, providing significantly faster results compared to traditional culture methods that can take 7-14 days.
-
Q: Is technical support available for setup and troubleshooting?
A: Yes, Cowingene provides comprehensive technical support, including detailed protocols, online resources, and direct assistance from our expert team, to ensure seamless integration and optimal performance of our kits in your laboratory.
Conclusion: The Future of Legionella Pneumophila Detection is Here
The ongoing battle against Legionella pneumophila demands vigilant monitoring and rapid, accurate detection solutions. As industries and public health bodies continue to prioritize proactive risk management, the role of advanced molecular diagnostics, particularly PCR-based kits, becomes increasingly central. The Cowingene Legionella Pneumophila Detection Kit (Liquid) stands at the forefront of this evolution, offering a robust, efficient, and reliable method for identifying and quantifying the presence of this critical pathogen. By significantly reducing turnaround times and providing superior sensitivity and specificity compared to traditional methods, our kit empowers B2B clients across healthcare, hospitality, industrial, and municipal sectors to implement effective water safety plans, respond swiftly to potential contamination, and ensure regulatory compliance. Our unwavering commitment to standards—demonstrated through scientific expertise, extensive field experience, authoritative validations, and unwavering trustworthiness—positions Cowingene as a reliable and strategic partner in your endeavors to safeguard public health. Embracing such cutting-edge solutions is not merely about adherence to guidelines; it is about investing in a safer, more resilient future. We invite organizations to leverage the power of advanced legionella pneumophila detection to enhance their environmental monitoring capabilities and protect their communities, employees, and customers from the significant risks associated with Legionella exposure. The comprehensive benefits, ranging from operational efficiency and cost savings to heightened safety and peace of mind, make the choice for rapid and reliable detection clear.
References:
- Centers for Disease Control and Prevention. (2022). Legionella (Legionnaires' Disease and Pontiac Fever). Retrieved from CDC website.
- European Centre for Disease Prevention and Control. (2017). European Guidelines for Legionella infection prevention and control in healthcare facilities. ECDC Technical Report.
- ASHRAE Standard 188-2018. (2018). Legionellosis: Risk Management for Building Water Systems. American Society of Heating, Refrigerating and Air-Conditioning Engineers.
- Whiley, H., & Bentham, R. (2011). Legionella and Legionnaires' Disease: A Review of Detection Methods, Prevention, and Control. Critical Reviews in Environmental Science and Technology, 41(21), 1957-1982.
- Public Health England. (2013). Guidance on the control of Legionella in hot and cold water systems. Health and Safety Executive (L8).
Related PRODUCTS
-
Comprehensive Stool GI Panel Analysis for Gut Health and Research Applications
NewsJan.27,2026 -
Understanding PCR DNA Stool Tests for Accurate Gastrointestinal Health Diagnostics
NewsJan.27,2026 -
PCR Diarrhea Panel: Fast, Accurate Gastrointestinal Pathogen Detection
NewsJan.06,2026 -
Comprehensive Guide to Diarrhea Pcr Panel for B2B Decision Makers | Cowingene
NewsJan.06,2026 -
Reliable Diarrhea PCR Kits for Fast Pathogen Detection | COWIN GENE
NewsJan.06,2026 -
Comprehensive Guide to Gi Pathogen Test Solutions for B2B Professionals
NewsJan.05,2026








