Aug . 26, 2025 05:20 Back to list
Expert Respiratory Panel Lab Services for Rapid Diagnosis
Navigating the Complexities of Respiratory Pathogen Diagnostics with Advanced Panel Solutions
In the rapidly evolving landscape of clinical diagnostics, the ability to accurately and swiftly identify respiratory pathogens is paramount for effective patient management, infection control, and public health surveillance. A comprehensive respiratory panel lab solution offers clinicians the power to differentiate between various viral and bacterial causes of acute respiratory infections, often presenting with overlapping symptoms. This article delves into the critical aspects of modern respiratory pathogen panels, covering their technical underpinnings, application versatility, industry trends, and the compelling advantages they offer to B2B stakeholders in healthcare.
The deployment of an advanced **respiratory panel lab** minimizes diagnostic ambiguity, reduces the reliance on empirical treatment, and plays a crucial role in combating antimicrobial resistance by enabling targeted therapeutic interventions. As global health challenges continue to underscore the importance of rapid diagnostics, the demand for robust and reliable **respiratory pathogen panel** technologies continues to escalate.
The Advanced Development Process of a Respiratory Panel Kit
Developing a high-performance **respiratory panel lab** kit is a meticulous process, demanding stringent quality control and adherence to international standards. The journey from conceptualization to market-ready product involves several critical stages, ensuring reliability, accuracy, and ease of use in diverse clinical settings.
Detailed Process Flow:
- 1. Pathogen Target Selection & Assay Design: Initial phase involves comprehensive epidemiological analysis to identify prevalent respiratory viruses and bacteria. This informs the selection of highly specific genetic targets for each pathogen. Oligonucleotide primers and probes are designed using advanced bioinformatics tools to ensure optimal sensitivity and specificity, avoiding cross-reactivity with commensal flora or other pathogens.
- 2. Reagent Formulation & Optimization: Essential components such as enzymes (e.g., reverse transcriptase, DNA polymerase), dNTPs, and reaction buffers are precisely formulated. Multiplexing strategies are developed to detect multiple targets simultaneously within a single reaction tube, requiring careful balancing of reagent concentrations to prevent assay interference and ensure uniform performance across all targets.
- 3. Manufacturing & Quality Control: Manufacturing processes adhere to Good Manufacturing Practices (GMP) and ISO 13485 standards. This includes automated pipetting for consistent reagent dispensing, lyophilization for extended shelf-life, and meticulous packaging. Each batch undergoes rigorous quality control testing for purity, concentration, activity, and freedom from contamination.
-
4. Analytical Validation: This stage confirms the kit's performance characteristics. Key parameters include:
- Limit of Detection (LoD): Determining the lowest concentration of pathogen that can be reliably detected.
- Analytical Specificity: Ensuring the assay does not react with non-target organisms or human genomic DNA.
- Linearity: Assessing the proportional relationship between target concentration and signal intensity across a range.
- Precision: Evaluating reproducibility across different runs, operators, and instruments.
- 5. Clinical Validation & Regulatory Approval: Clinical samples are tested to assess the kit's performance against reference methods, yielding data on diagnostic sensitivity and specificity. Regulatory bodies like the FDA, CE IVD, or local health authorities review extensive data packages to grant market approval, ensuring the kit meets stringent safety and efficacy requirements.
The typical service life of these kits, when stored appropriately, ranges from 12 to 24 months, ensuring long-term usability for target industries including clinical diagnostic laboratories, public health institutions, large hospital networks, and reference labs. The primary advantage in application scenarios, such as rapidly differentiating between influenza, RSV, and bacterial pneumonia, lies in enabling precise patient care and preventing unnecessary antibiotic use.
Industry Trends and the Evolution of Respiratory Diagnostics
The global diagnostics market for respiratory infections is experiencing unprecedented growth, driven by several key trends. The increasing prevalence of respiratory diseases, the continuous emergence of novel pathogens, and the critical lessons learned from recent pandemics have accelerated the adoption of advanced molecular diagnostic technologies.
- Shift Towards Syndromic Testing: Instead of testing for individual pathogens, syndromic panels allow simultaneous detection of multiple viral and bacterial targets from a single sample. This comprehensive approach is crucial for diseases with overlapping symptoms, saving time and resources.
- Automation and Integration: Laboratories are increasingly seeking automated solutions for sample preparation, nucleic acid extraction, amplification, and result interpretation. This reduces hands-on time, minimizes human error, and improves laboratory throughput, essential for high-volume testing centers.
- Point-of-Care (POC) Diagnostics: While high-throughput central lab panels remain vital, there's a growing interest in rapid, decentralized POC **respiratory panel test for** immediate results, particularly in emergency departments or remote settings.
- Focus on Specific and Atypical Pathogens: Beyond common viruses, panels are expanding to include less common but clinically significant pathogens like *Legionella pneumophila*, *Mycoplasma pneumoniae*, and *Chlamydophila pneumoniae*. For example, the Cowingene Legionella Pneumophila Detection Kit (NATBox) offers a highly sensitive and specific molecular solution for this critical atypical bacterial pathogen, complementing broader panels or serving as a confirmatory test when Legionellosis is suspected.
- Data Connectivity and LIS Integration: Seamless integration with Laboratory Information Systems (LIS) is a critical trend, enabling efficient data management, result reporting, and epidemiological tracking.
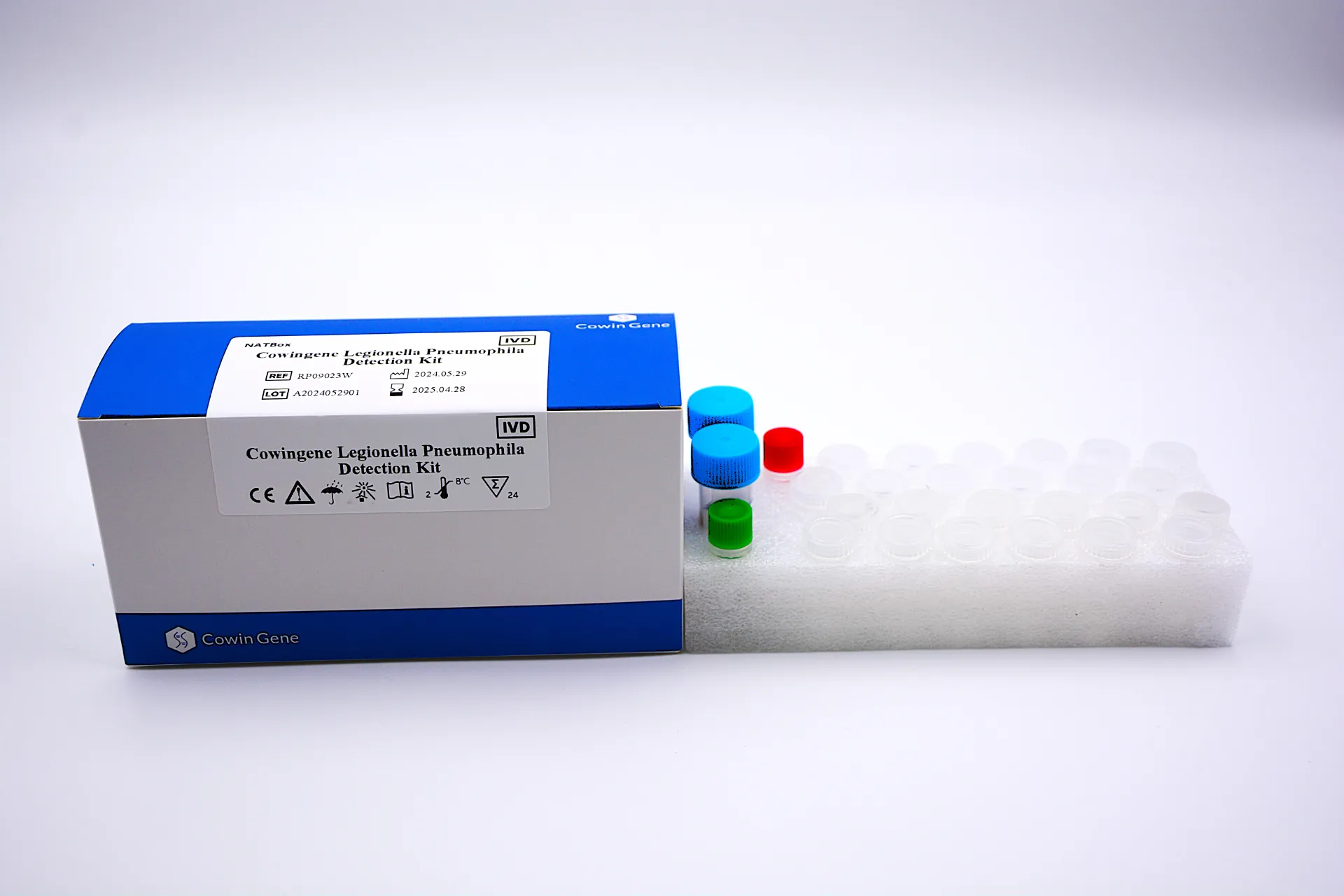
Figure 1: Illustration of a modern molecular diagnostic workflow for respiratory panels.
Technical Specifications and Key Parameters
The performance of a **respiratory panel lab** is defined by its technical specifications, which directly impact diagnostic accuracy and clinical utility. Key parameters such as sensitivity, specificity, and turnaround time are crucial for evaluating the efficacy of any molecular diagnostic platform.
| Parameter | Description/Typical Value Range |
|---|---|
| Targets Detected | 15-25 common respiratory viruses and bacteria (e.g., Influenza A/B, RSV, SARS-CoV-2, Adenovirus, Parainfluenza, Metapneumovirus, Rhinovirus/Enterovirus, *Mycoplasma pneumoniae*, *Chlamydia pneumoniae*, *Bordetella pertussis*). Specific kits like Cowingene's target *Legionella pneumophila* alone for focused detection. |
| Sample Type | Nasopharyngeal swabs, oropharyngeal swabs, bronchoalveolar lavage (BAL), sputum. |
| Limit of Detection (LoD) | Typically 100-1000 copies/mL or 10-100 TCID50/mL, depending on the pathogen and assay. For Cowingene's Legionella kit, LoD is optimized for highly sensitive detection. |
| Clinical Sensitivity | >95% (compared to gold standard methods). |
| Clinical Specificity | >98% (ensuring minimal false positives). |
| Turnaround Time (TAT) | 1-3 hours (instrument run time), total workflow can be 3-5 hours depending on sample processing and automation level. |
| Regulatory Status | CE-IVD marked, FDA EUA (Emergency Use Authorization) or 510(k) cleared, or other relevant regional approvals. |
Application Scenarios and Technical Advantages
The versatility of a modern **respiratory pathogen panel** extends across various clinical and public health settings, providing distinct technical and operational advantages.
Key Application Scenarios:
- Emergency Departments & Critical Care: Rapid identification of causative agents allows for timely initiation of targeted antiviral or antibacterial therapy, reducing hospital stays and improving patient outcomes, especially for severe cases like pneumonia or ARDS.
- Infection Control & Epidemiology: Quick and accurate diagnosis helps in isolating infected individuals and tracking outbreaks, preventing nosocomial transmission within healthcare facilities and facilitating public health interventions.
- Immunocompromised Patients: For patients with weakened immune systems (e.g., transplant recipients, oncology patients), a prompt diagnosis is critical as respiratory infections can be particularly severe and rapidly progressive.
- Antibiotic Stewardship Programs: By differentiating between viral and bacterial infections, these panels guide appropriate antibiotic use, reducing unnecessary prescriptions and combating the rise of antimicrobial resistance.
- Outpatient Clinics & Primary Care: Enabling precise diagnosis, a **respiratory panel test for** common infections streamlines patient flow and reduces follow-up visits.
Technical Advantages:
- Multiplexing Capabilities: Detecting multiple targets simultaneously from a single patient sample, saving time and sample volume compared to serial single-target tests.
- High Sensitivity and Specificity: Molecular methods offer superior analytical performance compared to traditional culture or antigen-based tests, particularly for fastidious organisms or low viral loads.
- Reduced Manual Error: Automated systems integrating sample preparation to result interpretation minimize human intervention and potential errors, ensuring consistent and reliable results.
- Scalability: Platforms can be scaled to meet varying testing demands, from individual tests to large-batch processing, crucial during seasonal peaks or public health crises.
- Comprehensive Coverage: Modern panels cover a broad spectrum of clinically relevant pathogens, including those that cause severe disease, such as the *Legionella pneumophila* detected by Cowingene's NATBox kit.

Figure 2: Schematic demonstrating the broad diagnostic capability of a respiratory panel.
Vendor Comparison and Customized Solutions
The market for **respiratory pathogen panel** solutions is competitive, with numerous vendors offering diverse platforms and target pathogen configurations. Selecting the right solution involves a thorough comparison of key features, performance, and vendor support.
Comparative Analysis:
| Feature | Vendor A (Broad Panel) | Vendor B (Medium Panel) | Cowingene (Focused, e.g., Legionella) |
|---|---|---|---|
| Number of Targets | 20+ (Viruses & Bacteria) | 10-15 (Common Viruses) | 1 (e.g., *Legionella pneumophila* - highly specific) |
| Platform Compatibility | Proprietary integrated system | Open RT-PCR system compatible | NATBox System (closed, integrated) |
| Turnaround Time | ~2-3 hours | ~1.5-2 hours | < 90 minutes (Cowingene NATBox) |
| Automation Level | High (sample-to-result) | Moderate (manual extraction) | High (integrated extraction & amplification) |
| Regulatory Approvals | CE-IVD, FDA 510(k) | CE-IVD, research use only (RUO) versions | CE-IVD (Cowingene Legionella Pneumophila Detection Kit (NATBox)) |
Cowingene, with its specialized solutions like the Legionella Pneumophila Detection Kit (NATBox), represents a vendor focusing on high-precision, rapid detection for specific, critical pathogens. While not a broad **respiratory panel lab** in itself, such focused kits are vital components within a comprehensive diagnostic strategy, enabling rapid, targeted testing when specific threats are identified or highly suspected.
Customized Solutions:
Recognizing that every laboratory has unique needs, many vendors, including Cowingene, offer customized solutions tailored to specific operational requirements:
- Panel Configuration: Tailoring the specific pathogens included in a panel based on regional epidemiology or clinical focus (e.g., adding endemic pathogens or removing less relevant ones).
- Instrument Integration: Adapting kits to run on existing laboratory equipment to minimize capital expenditure and training needs.
- LIS Interfacing: Developing custom interfaces for seamless data transfer and integration with a laboratory's existing information system for streamlined reporting.
- Throughput Optimization: Designing workflows and providing instrumentation to match required sample processing volumes, from low-throughput batch processing to high-throughput continuous operation.
Figure 3: Advanced molecular diagnostic platform for rapid pathogen detection.
Real-World Application Case Studies
The tangible benefits of implementing advanced **respiratory panel lab** solutions are best illustrated through real-world applications. These case studies highlight improvements in patient care, operational efficiency, and public health outcomes.
Case Study 1: Rapid Diagnosis in an Urban Hospital
An urban tertiary care hospital implemented a high-throughput **respiratory pathogen panel** for all patients admitted with acute respiratory symptoms. Prior to this, individual PCR tests or traditional cultures were used, leading to delays of 24-48 hours. With the new panel, results were available within 3-4 hours of sample receipt.
- Outcome: A 40% reduction in the average length of stay for patients with viral respiratory infections due to earlier discharge. A 25% decrease in empiric antibiotic use for viral cases, significantly contributing to the hospital’s antibiotic stewardship efforts.
- Customer Feedback: "The rapid turnaround of the respiratory panel revolutionized our emergency department workflow. We can now make informed treatment decisions within hours, which is critical for our patient population," commented the Head of Microbiology.
Case Study 2: Public Health Surveillance during a Seasonal Outbreak
During a severe influenza season, a regional public health laboratory utilized an advanced **respiratory panel test for** comprehensive pathogen detection to monitor circulating strains and detect co-infections. This allowed for detailed epidemiological tracking and proactive public health messaging.
- Outcome: Early identification of a novel co-infection pattern (e.g., Influenza A with *Mycoplasma pneumoniae*) allowed for targeted public health warnings and adjusted clinical guidelines for treatment. The rapid data helped predict surges in hospital admissions and allocate resources effectively.
- Authoritative Reference: Data from this surveillance was published in a regional public health journal, demonstrating the utility of the panel in real-time epidemic management.
Case Study 3: Addressing Atypical Pneumonia with Focused Diagnostics
A remote healthcare facility faced an cluster of severe pneumonia cases unresponsive to standard antibiotics. Initial broad **respiratory panel lab** results were inconclusive for common viral or bacterial agents. Prompt suspicion of an atypical pathogen led them to employ a specialized kit, such as the Cowingene Legionella Pneumophila Detection Kit (NATBox), for rapid and definitive diagnosis.
- Outcome: The Cowingene NATBox kit quickly and accurately identified *Legionella pneumophila* as the causative agent. This enabled immediate initiation of appropriate macrolide therapy, significantly improving patient prognosis and preventing further spread through environmental investigation.
- Experience: "Having a highly specific and rapid test for *Legionella* was a game-changer. It allowed us to pinpoint the problem quickly, saving lives and guiding critical public health interventions," stated the lead physician.
Ensuring Trust and Reliability: FAQs, Fulfillment, and Support
Trustworthiness is a cornerstone of B2B relationships, particularly in critical sectors like diagnostics. Providers of **respiratory panel lab** solutions must offer transparent information on product reliability, logistical support, and post-sales service.
Frequently Asked Questions (FAQs):
A1: Typical requirements range from 100-300 µL of sample (e.g., viral transport medium from a nasopharyngeal swab), though specific kits may vary. Refer to product inserts for precise details.
A2: Our assay designs target highly conserved regions of pathogen genomes, offering broad detection capabilities. Continuous surveillance and in silico analysis are performed to ensure efficacy against emerging variants. Updates are provided as necessary.
A3: Many of our panels are validated on common automated nucleic acid extraction platforms. Specific compatibility lists are available upon request to ensure seamless integration into your laboratory workflow.
Lead Time and Fulfillment:
Our standard lead time for most **respiratory panel lab** kits is 2-4 weeks from order confirmation, depending on volume and specific product configuration. For urgent requirements or large-scale procurements, we offer expedited shipping options and work closely with clients to manage logistics. Inventory levels are continuously monitored to ensure consistent supply, especially during seasonal demand peaks.
Warranty Commitments:
All our diagnostic kits are backed by a comprehensive warranty, guaranteeing performance within stated specifications when stored and used according to the provided instructions for the duration of their stated shelf life. This commitment reflects our confidence in the rigorous quality control applied during manufacturing (ISO 13485 certified processes).
Customer Support and Training:
We provide extensive customer support, including:
- Dedicated Technical Support: Available via phone and email for troubleshooting and assay-related queries.
- On-site & Remote Training: Comprehensive training programs for laboratory personnel on product usage, instrument operation, and data interpretation.
- Application Specialists: Experts available for consultation on workflow optimization and customization.
- Regular Software Updates: Ensuring optimal performance and compatibility with evolving LIS requirements.
Conclusion
The demand for precise and rapid respiratory pathogen diagnostics has never been higher. Modern **respiratory panel lab** solutions, whether broad syndromic assays or highly focused detection kits like the Cowingene Legionella Pneumophila Detection Kit (NATBox), offer unparalleled advantages in patient care, public health, and antimicrobial stewardship. By leveraging advanced molecular technologies, adhering to stringent quality standards, and providing robust support, diagnostic companies are empowering healthcare professionals to make critical decisions with confidence, ultimately improving health outcomes globally.
References
- Metzgar D, et al. Molecular epidemiology of respiratory viruses in hospitalized children. J Clin Microbiol. 2021;59(5):e02316-20.
- Perlman S. Coronavirus Infectious Bronchitis Virus, SARS, and MERS. Adv Virus Res. 2020;107:1-55.
- Centers for Disease Control and Prevention. Legionellosis (Legionnaires' Disease and Pontiac Fever). Available at: cdc.gov/legionella/. Accessed October 26, 2023.
- Pneumonia Etiology Research for Child Health (PERCH) Study Group. Causes of severe pneumonia in children younger than 5 years: a multicentre prospective cohort study. Lancet. 2019;394(10200):757-779.
- World Health Organization. Antimicrobial resistance. Available at: who.int/news-room/fact-sheets/detail/antimicrobial-resistance. Accessed October 26, 2023.
Related PRODUCTS
-
Comprehensive Stool GI Panel Analysis for Gut Health and Research Applications
NewsJan.27,2026 -
Understanding PCR DNA Stool Tests for Accurate Gastrointestinal Health Diagnostics
NewsJan.27,2026 -
PCR Diarrhea Panel: Fast, Accurate Gastrointestinal Pathogen Detection
NewsJan.06,2026 -
Comprehensive Guide to Diarrhea Pcr Panel for B2B Decision Makers | Cowingene
NewsJan.06,2026 -
Reliable Diarrhea PCR Kits for Fast Pathogen Detection | COWIN GENE
NewsJan.06,2026 -
Comprehensive Guide to Gi Pathogen Test Solutions for B2B Professionals
NewsJan.05,2026








