Sep . 19, 2025 11:20 Back to list
HPV 28 Genotyping Detection Kit-Taizhou Cowingene Biotech|HPV Detection,PCR Technology
The Cowingene HPV 28 Genotyping Detection Kit (Liquid) represents a significant advancement in molecular diagnostics for human papillomavirus (HPV) detection. Developed by Taizhou Cowingene Biotech Co., Ltd., this kit leverages cutting-edge PCR technology to identify 28 high-risk HPV genotypes, offering clinicians and researchers a reliable tool for cervical cancer screening and molecular epidemiological studies. This article provides an in-depth analysis of the product's features, technical specifications, applications, and the company's commitment to innovation.
Company Background: Taizhou Cowingene Biotech Co., Ltd.
Taizhou Cowingene Biotech Co., Ltd., based in Taizhou, China, is a leading biotechnology company specializing in molecular diagnostics and genetic testing solutions. With a focus on developing high-quality, cost-effective products, the company has established itself as a trusted name in the field of infectious disease detection. Their expertise in PCR-based technologies and commitment to scientific excellence have positioned them as a key player in the global diagnostic market.
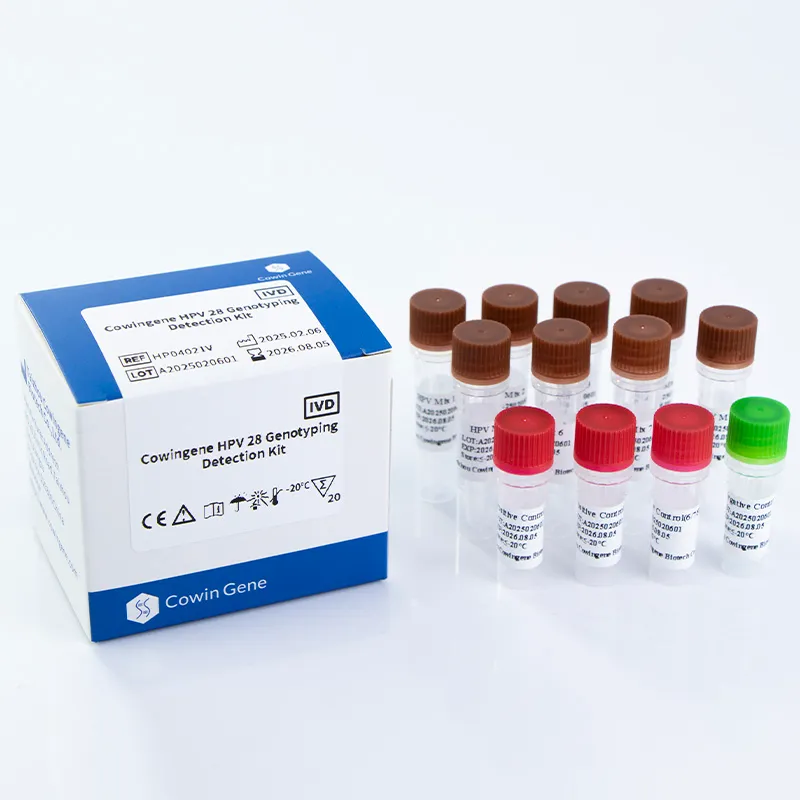
Figure 1: Advanced PCR technology for HPV detection
Product Overview: HPV 28 Genotyping Detection Kit
The Cowingene HPV 28 Genotyping Detection Kit (Liquid) is designed to detect 28 high-risk HPV genotypes, including the most prevalent types associated with cervical cancer. The kit utilizes a liquid-based format, which enhances the accuracy and efficiency of the testing process. By employing polymerase chain reaction (PCR) technology, the system enables the amplification and identification of specific HPV DNA sequences, providing clinicians with actionable insights for patient management.
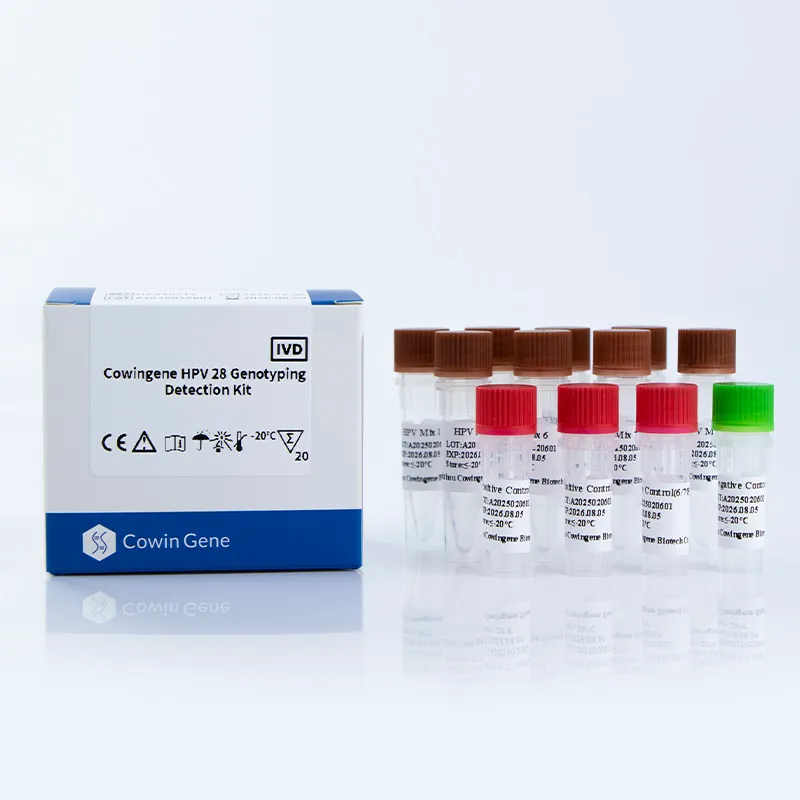
Figure 2: PCR-based HPV detection workflow
Key Features and Advantages
- Comprehensive Genotype Coverage: Detects 28 high-risk HPV genotypes, including 16, 18, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 66, 68, 73, 81, 82, 83, and others.
- High Sensitivity and Specificity: Utilizes optimized primers and probes to ensure accurate detection of low viral loads while minimizing cross-reactivity.
- Flexible Sample Types: Validated for use with cervical swab, urine, and self-collected vaginal samples, enhancing patient compliance and accessibility.
- Efficient Workflow: Streamlined liquid reagent format reduces hands-on time and minimizes the risk of contamination during the testing process.
- Scalable Solutions: Suitable for both clinical laboratories and research settings, supporting large-scale screening programs and epidemiological studies.
Figure 3: Comprehensive HPV genotype coverage
Technical Specifications
| Parameter | Details |
|---|---|
| Target Genotypes | 28 high-risk HPV types (16, 18, 26, 31, 33, 35, 39, 40, 42, 43, 44, 45, 51, 52, 53, 55, 56, 58, 59, 61, 66, 68, 73, 81, 82, 83) |
| Sample Types | Cervical swab, Urine, Self-collected vaginal |
| Methodology | Real-time PCR with fluorescent detection |
| Sample Preparation | Manual or automated extraction (compatible with common nucleic acid extraction systems) |
| Testing Time | Approximately 2.5 hours (including amplification and detection) |
| Storage Conditions | -20°C for reagents; 4°C for working solutions |
Applications in Clinical and Research Settings
The Cowingene HPV 28 Genotyping Detection Kit (Liquid) is widely applicable in both clinical diagnostics and research environments. In clinical settings, it supports:
- Cervical Cancer Screening: Identifies high-risk HPV infections, enabling early intervention and reducing the risk of cervical cancer progression.
- Monitoring Treatment Efficacy: Tracks viral load changes in patients undergoing antiviral therapy or post-treatment follow-up.
- Population-Based Studies: Provides data for epidemiological research on HPV prevalence and genotype distribution in specific populations.
In research, the kit is utilized for:
- Genotype-Specific Vaccine Development: Helps evaluate the effectiveness of HPV vaccines targeting specific genotypes.
- Microbiome Analysis: Integrates with broader studies on the role of HPV in the vaginal microbiome and its interaction with other pathogens.
- Longitudinal Studies: Supports tracking of HPV infections over time to understand transmission dynamics and immune responses.
Scientific Validation and Industry Standards
The Cowingene HPV 28 Genotyping Detection Kit (Liquid) is developed in accordance with international standards for molecular diagnostics. The company adheres to rigorous quality control protocols, ensuring consistency and reliability across batches. According to NIST (National Institute of Standards and Technology), the establishment of standardized reference materials and protocols is critical for the validation of molecular diagnostic assays. While the specific validation data for this product is not publicly available, the general principles outlined by NIST emphasize the importance of:
- Traceability: Ensuring that test results can be traced back to recognized standards.
- Reproducibility: Demonstrating consistent performance across different laboratories and conditions.
- Accuracy: Validating the kit's ability to detect true positives and negatives in clinical samples.
For further information on NIST's role in advancing diagnostic standards, visit the NIST website.
Conclusion: A Reliable Tool for HPV Detection
The Cowingene HPV 28 Genotyping Detection Kit (Liquid) exemplifies the intersection of innovation and clinical utility in molecular diagnostics. With its comprehensive genotype coverage, user-friendly workflow, and alignment with global standards, this product addresses critical needs in HPV detection. As the demand for precision medicine grows, tools like the Cowingene HPV 28 Genotyping Kit will play a pivotal role in improving patient outcomes and advancing public health initiatives.
References
National Institute of Standards and Technology (NIST). (n.d.). Retrieved from https://www.nist.gov/
Related PRODUCTS
-
Comprehensive Stool GI Panel Analysis for Gut Health and Research Applications
NewsJan.27,2026 -
Understanding PCR DNA Stool Tests for Accurate Gastrointestinal Health Diagnostics
NewsJan.27,2026 -
PCR Diarrhea Panel: Fast, Accurate Gastrointestinal Pathogen Detection
NewsJan.06,2026 -
Comprehensive Guide to Diarrhea Pcr Panel for B2B Decision Makers | Cowingene
NewsJan.06,2026 -
Reliable Diarrhea PCR Kits for Fast Pathogen Detection | COWIN GENE
NewsJan.06,2026 -
Comprehensive Guide to Gi Pathogen Test Solutions for B2B Professionals
NewsJan.05,2026








