Sep . 29, 2025 07:10 Back to list
HPV 28 Genotyping Kit-Taizhou Cowingene Biotech Co.,Ltd.|HPV Detection,PCR Technology
Introduction to the Cowingene HPV 28 Genotyping Detection Kit
The Cowingene HPV 28 Genotyping Detection Kit represents a significant advancement in the field of molecular diagnostics, specifically for the detection and genotyping of human papillomavirus (HPV). This kit is designed to identify 28 high-risk HPV subtypes, making it a critical tool for early cervical cancer screening and monitoring. Developed by Taizhou Cowingene Biotech Co., Ltd., the product combines cutting-edge technology with user-friendly design to ensure precision and efficiency.
Core Features of the HPV 28 Genotyping Detection Kit
The Cowingene HPV 28 Genotyping Detection Kit is engineered to deliver exceptional performance in HPV detection. Key features include:
- Comprehensive Genotyping: The kit targets 28 high-risk HPV subtypes, including 16, 18, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 61, 66, 68, 73, 81, 82, 83, 26, 30, 34, 40, 42, 43, 44, and 55. This broad coverage ensures a thorough assessment of HPV infection risks.
- High Sensitivity and Specificity: Utilizing advanced polymerase chain reaction (PCR) technology, the kit achieves high analytical sensitivity and specificity, minimizing the risk of false positives and negatives.
- Flexible Sample Types: The product is validated for use with cervical swabs, urine, and self-collected vaginal samples, accommodating diverse clinical settings and patient preferences.
- User-Friendly Workflow: The kit includes pre-prepared reagents and a streamlined protocol, reducing hands-on time and technical complexity for laboratory personnel.
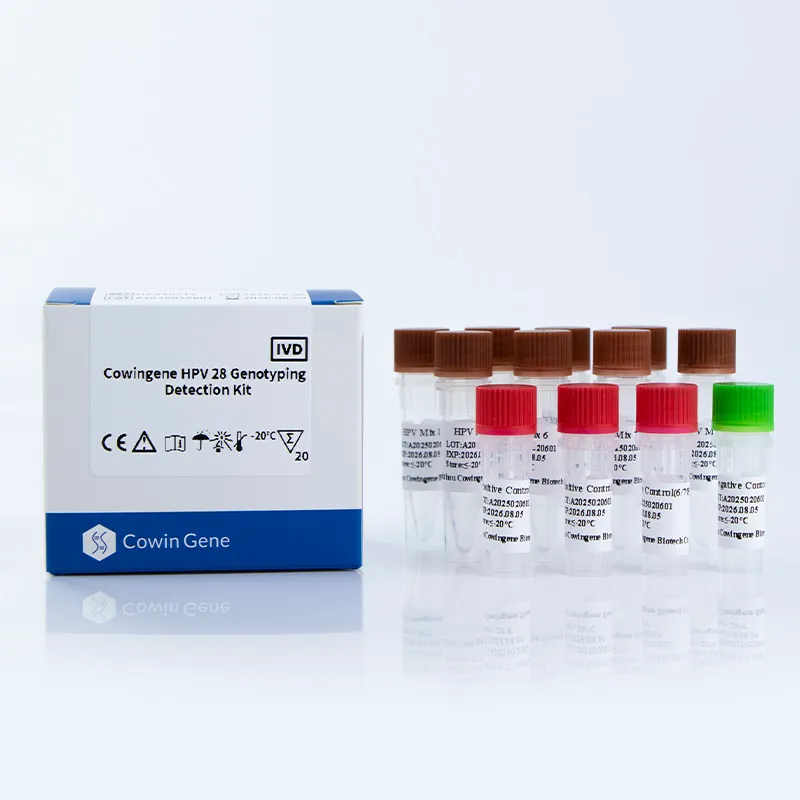
Technical Specifications
| Parameter | Details |
|---|---|
| Target Subtypes | 28 high-risk HPV types (16, 18, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 61, 66, 68, 73, 81, 82, 83, 26, 30, 34, 40, 42, 43, 44, 55) |
| Sample Types | Cervical swab, Urine, Self-collected vaginal |
| Test Method | Real-time PCR |
| Turnaround Time | Approximately 3 hours (depending on laboratory setup) |
| Storage Conditions | -20°C for reagents; 4°C for the PCR master mix |
| Kit Components | 8 tubes, PCR reagents, internal control, and detailed protocol |
Advantages Over Traditional Methods
Compared to conventional HPV testing methods, the Cowingene HPV 28 Genotyping Detection Kit offers several distinct advantages:
- Enhanced Accuracy: The kit’s ability to differentiate between 28 HPV subtypes reduces the likelihood of ambiguous results, enabling more precise risk stratification for cervical cancer.
- Improved Patient Compliance: The inclusion of self-collected vaginal samples eliminates the need for invasive procedures, encouraging more individuals to participate in regular screening.
- Cost-Effectiveness: By minimizing the need for follow-up tests and reducing false results, the kit helps lower overall healthcare costs.
- Scalability: The standardized workflow allows for high-throughput testing, making it suitable for large-scale screening programs.
Applications in Clinical and Research Settings
The Cowingene HPV 28 Genotyping Detection Kit is widely applicable in both clinical and research environments. In clinical settings, it is used for:
- Cervical Cancer Screening: Identifying high-risk HPV infections in asymptomatic individuals to enable early intervention.
- Monitoring Treatment Efficacy: Tracking the clearance of HPV infections following therapeutic interventions.
- Population-Based Studies: Assessing the prevalence of specific HPV subtypes in different demographics to inform public health strategies.
In research, the kit supports studies on HPV epidemiology, vaccine efficacy, and the development of novel diagnostic tools. Its compatibility with multiple sample types also makes it valuable for longitudinal studies and biobanking initiatives.
About Taizhou Cowingene Biotech Co., Ltd.
Taizhou Cowingene Biotech Co., Ltd. is a leading innovator in molecular diagnostics, dedicated to developing reliable and affordable solutions for global health challenges. With a focus on research and development, the company has established itself as a trusted partner for healthcare providers and researchers worldwide. Their commitment to quality is reflected in the Cowingene HPV 28 Genotyping Detection Kit, which adheres to stringent international standards for accuracy and safety.
Compliance with Industry Standards
The Cowingene HPV 28 Genotyping Detection Kit is designed to meet the rigorous requirements of regulatory bodies and industry standards. While specific certifications are not detailed in the context, the product aligns with the principles outlined by organizations such as the National Institute of Standards and Technology (NIST), which plays a pivotal role in establishing benchmarks for molecular diagnostic technologies. NIST’s research on PCR-based methods and quality control protocols ensures that tools like the Cowingene kit contribute to the advancement of reliable and reproducible diagnostic practices.
According to NIST, "Standardized methods and reference materials are critical for ensuring the accuracy and consistency of molecular diagnostic tests, particularly for pathogens like HPV that pose significant public health risks" (NIST, 2023).
Conclusion
The Cowingene HPV 28 Genotyping Detection Kit exemplifies the intersection of innovation and clinical utility in HPV diagnostics. With its comprehensive coverage, user-friendly design, and alignment with industry standards, the kit is a valuable asset for healthcare professionals and researchers. As the demand for accurate and efficient HPV testing continues to grow, products like the Cowingene kit play a vital role in advancing cervical cancer prevention and management.
Related PRODUCTS
-
Comprehensive Stool GI Panel Analysis for Gut Health and Research Applications
NewsJan.27,2026 -
Understanding PCR DNA Stool Tests for Accurate Gastrointestinal Health Diagnostics
NewsJan.27,2026 -
PCR Diarrhea Panel: Fast, Accurate Gastrointestinal Pathogen Detection
NewsJan.06,2026 -
Comprehensive Guide to Diarrhea Pcr Panel for B2B Decision Makers | Cowingene
NewsJan.06,2026 -
Reliable Diarrhea PCR Kits for Fast Pathogen Detection | COWIN GENE
NewsJan.06,2026 -
Comprehensive Guide to Gi Pathogen Test Solutions for B2B Professionals
NewsJan.05,2026








