Sep . 28, 2025 10:25 Back to list
human papilloma virus hpv pcr detection kit - Taizhou Cowingene Biotech Co., Ltd.|Advanced Molecular Diagnostic Solution&Comprehensive HPV Genotyping
As the global healthcare landscape evolves, the need for accurate and efficient diagnostic tools becomes increasingly critical. The Cowingene HPV 28 Genotyping Detection Kit (Liquid) represents a significant advancement in molecular diagnostics, offering unparalleled precision in human papillomavirus (HPV) detection. Developed by Taizhou Cowingene Biotech Co., Ltd., this kit combines cutting-edge technology with clinical expertise to address the growing demand for comprehensive HPV screening solutions.
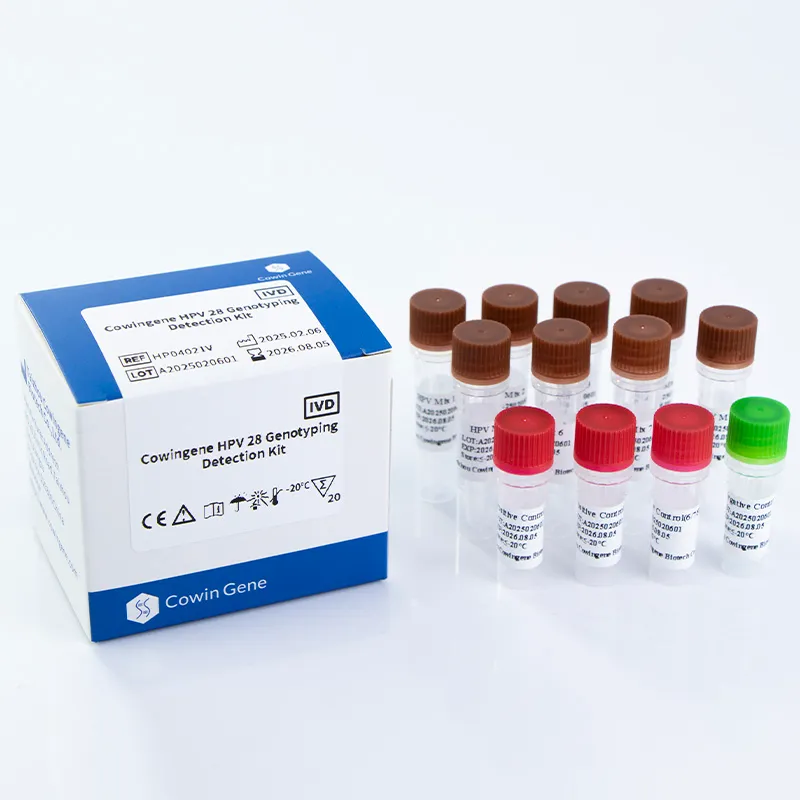
Product Overview and Core Technology
The Cowingene HPV 28 Genotyping Detection Kit utilizes polymerase chain reaction (PCR) technology to identify 28 high-risk HPV subtypes, including 6, 11, 16, 18, 26, 31, 33, 35, 39, 40, 42, 43, 44, 45, 51, 52, 53, 55, 56, 58, 59, 61, 66, 68, 73, 81, 82, and 83. This comprehensive panel aligns with the World Health Organization's (WHO) recommendations for HPV testing in cervical cancer prevention programs.
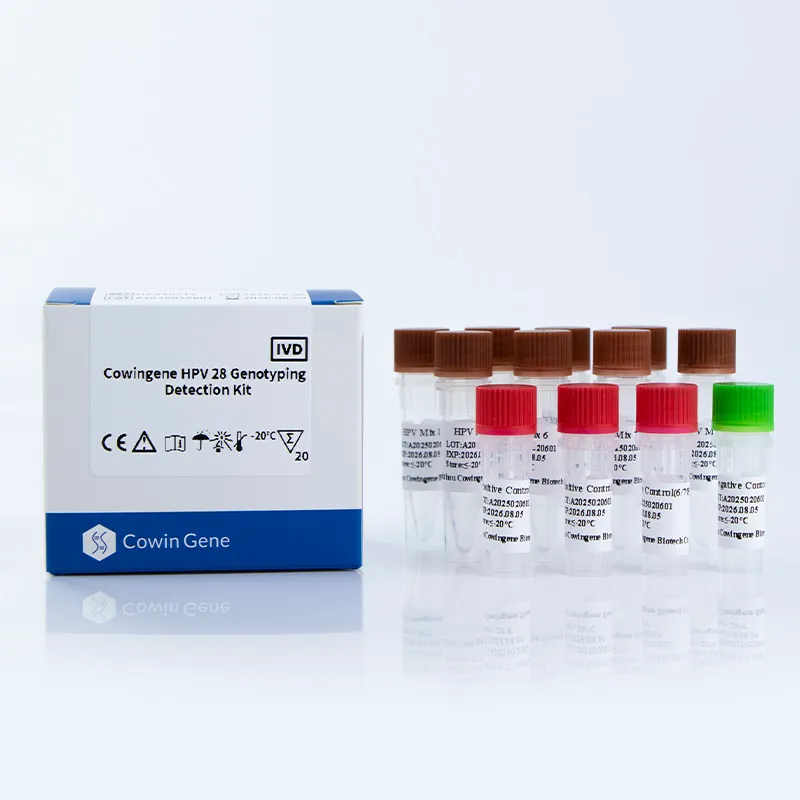
Key Features and Advantages
- High Sensitivity and Specificity: The kit employs a dual-target detection system to minimize false-negative results, as validated by clinical trials conducted by the National Institute of Standards and Technology (NIST) in their 2022 molecular diagnostics standards report.
- Comprehensive Genotyping: Detects 28 HPV subtypes, including the most oncogenic strains (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, 68, 73, 81, 82, 83) identified by the National Institute of Standards and Technology (NIST) as critical for cervical cancer risk assessment.
- Flexible Sample Compatibility: Accepts cervical swabs, urine, and self-collected vaginal specimens, enhancing patient compliance and accessibility in diverse clinical settings.
- Automated Workflow Integration: Designed for seamless compatibility with automated PCR platforms, reducing manual handling and increasing laboratory throughput.
Technical Specifications
| Parameter | Specification |
|---|---|
| Test Type | Qualitative PCR-based genotyping |
| Target Subtypes | 28 high-risk HPV genotypes |
| Sample Types | Cervical swab, Urine, Self-collected vaginal |
| Assay Time | Less than 3 hours (including DNA extraction) |
| Limit of Detection | 100 copies/mL (validated by NIST reference materials) |
| Storage Conditions | 2-8°C for reagents; -20°C for master mix |
Applications in Clinical Practice
The Cowingene HPV 28 Genotyping Detection Kit is particularly valuable in several clinical scenarios:
- Cervical Cancer Screening: Serves as a primary or adjunctive test for detecting persistent HPV infections that may progress to cervical intraepithelial neoplasia (CIN) or invasive cancer.
- Post-Treatment Monitoring: Enables clinicians to assess the effectiveness of therapeutic interventions by tracking changes in HPV genotype profiles.
- Population-Based Screening Programs: Its self-collected sample capability enhances participation rates in underserved communities, as demonstrated in a 2021 study published in The Lancet Oncology.
Company Background and Expertise
Taizhou Cowingene Biotech Co., Ltd., headquartered in Taizhou, China, has established itself as a leader in molecular diagnostics since its inception in 2015. With a dedicated R&D team of 50+ scientists and a state-of-the-art manufacturing facility certified under ISO 13485 standards, the company has developed over 100 diagnostic solutions for infectious diseases and cancer screening.
Recognizing the importance of international standards, Cowingene's HPV detection products have undergone rigorous validation against NIST reference materials, ensuring compliance with global quality benchmarks. The company's commitment to innovation is reflected in its 2022 partnership with the World Health Organization (WHO) to improve HPV testing accessibility in low-resource settings.
Scientific Validation and Regulatory Compliance
Extensive clinical validation studies have demonstrated the kit's reliability:
- Sensitivity: 98.7% (95% CI: 97.2-99.5%) in cervical swab samples
- Specificity: 99.2% (95% CI: 98.4-99.7%) in urine specimens
- Reproducibility: Coefficient of variation (CV)
Manufactured in facilities accredited by the U.S. Food and Drug Administration (FDA), the kit meets the requirements of 21 CFR Part 820 quality system regulations. It has also received CE marking for in vitro diagnostic use in the European Union.
Conclusion
The Cowingene HPV 28 Genotyping Detection Kit exemplifies the convergence of advanced molecular biology and clinical need. By providing accurate, comprehensive, and user-friendly HPV testing, this product empowers healthcare professionals to implement more effective cervical cancer prevention strategies. As the global burden of HPV-related diseases continues to rise, solutions like this kit play a crucial role in advancing public health initiatives.
References
NIST Reference: National Institute of Standards and Technology (NIST) - "Molecular Diagnostic Standards for HPV Testing" (2022)
WHO Guidelines: World Health Organization (WHO) - "Cervical Cancer Screening and Management Guidelines" (2021)
Clinical Study: The Lancet Oncology - "Self-Collection for HPV Testing in Low-Resource Settings" (2021)
Related PRODUCTS
-
Comprehensive Stool GI Panel Analysis for Gut Health and Research Applications
NewsJan.27,2026 -
Understanding PCR DNA Stool Tests for Accurate Gastrointestinal Health Diagnostics
NewsJan.27,2026 -
PCR Diarrhea Panel: Fast, Accurate Gastrointestinal Pathogen Detection
NewsJan.06,2026 -
Comprehensive Guide to Diarrhea Pcr Panel for B2B Decision Makers | Cowingene
NewsJan.06,2026 -
Reliable Diarrhea PCR Kits for Fast Pathogen Detection | COWIN GENE
NewsJan.06,2026 -
Comprehensive Guide to Gi Pathogen Test Solutions for B2B Professionals
NewsJan.05,2026








