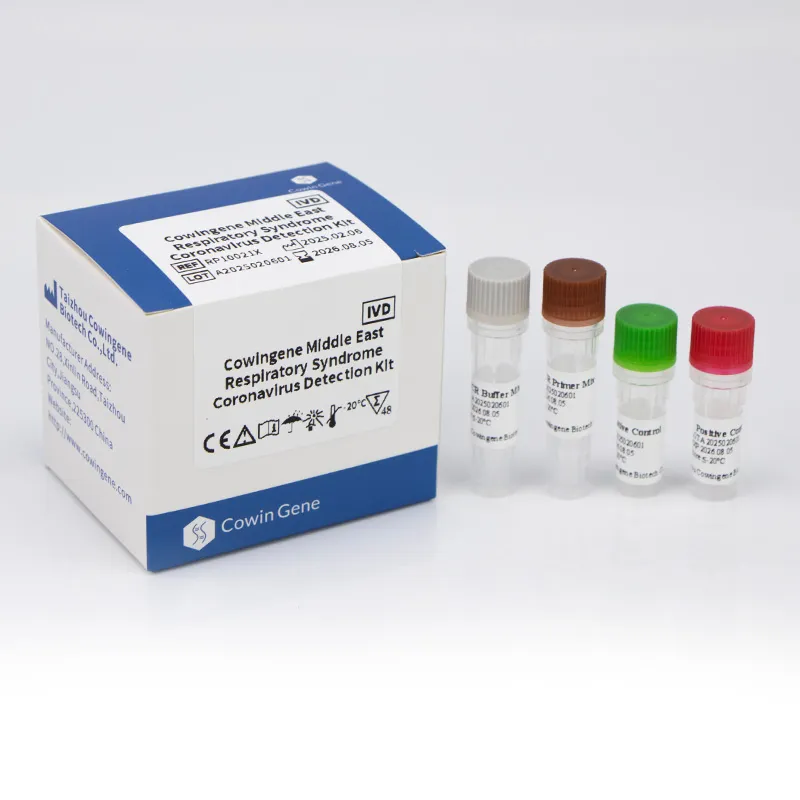
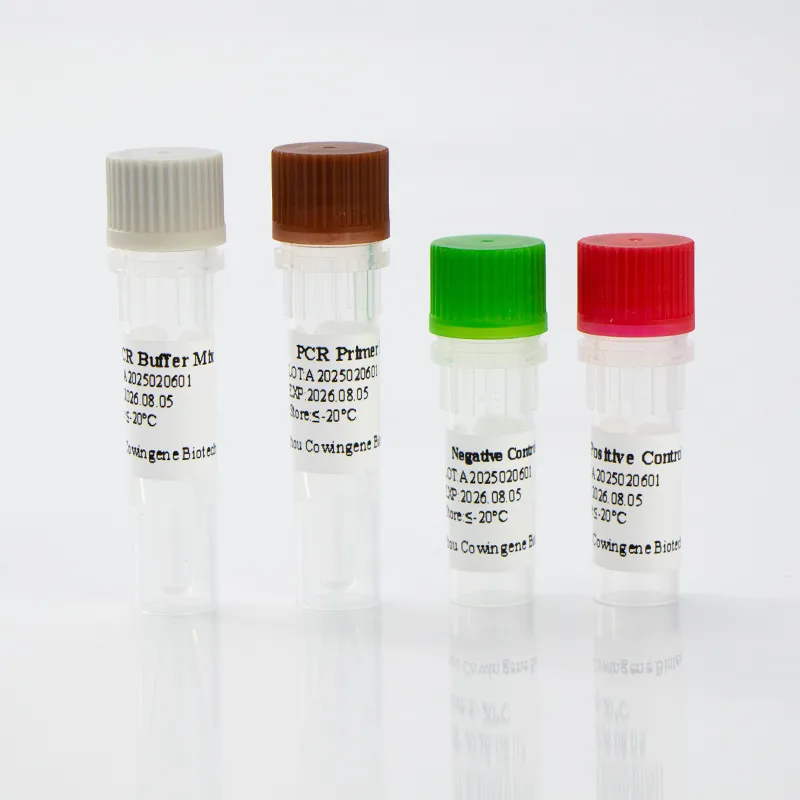
Cowingene Middle East Respiratory Syndrome Coronavirus Detection Kit (Liquid)
Intented Use
The Cowingene Middle East Respiratory Syndrome Coronavirus Detection Kit (MERS Detection Kit) is an in vitro diagnostic test for the qualitative detection of RNA from Middle East Respiratory Syndrome Coronavirus (MERS) in patient specimens. Including nasopharyngeal swab, nasopharyngeal aspirate, bronchoalveolar lavage, throat swab. For sample collection, it is recommended to refer to the laboratory technical guide.
Order Information
| REF |
Product Name |
Type |
Package Size |
| RP10021X |
Cowingene Middle East Respiratory Syndrome Coronavirus Detection Kit |
Liquid |
48 tests/kit |
Specification
Detection Target
Middle East Respiratory Syndrome Coronavirus
Storage
≤-20℃ for 18 months
Validated Specimen
Nasopharyngeal swab, Nasopharyngeal aspirate, Bronchoalveolar lavage, Throat swab
Compatible Instruments
Open real time PCR machine with channels FAM, VIC/HEX, ROX, CY5, i.e. ABI 7500, Roche 480, Bio-Rad CFX96, etc.
Storage and Handling Requirements
A. Reagents can be stored at≤-20℃ for 18 months
B. Reagents can be transported at≤-20℃ for no more than 15 days
C. Do not use reagents past expiration date indicated on outside of package
D. Protect from light
E. No freeze-thawing for more than 5 cycles
F. Once opened, reagent is valid for 7 days at 2-8℃
G. Prior to use, remove reagents from freezer and allow them to thaw at least 30 minutes at room temperature
H. Vortex reagents prior to each use
I. Do not store in frost free freezer
Recommended Supporting Reagents
| REF |
Product Name |
Certification |
Package Size |
| CW01021Y |
Cowingene Viral DNA/RNA Column |
CE |
50 tests/kit |
| CW02021Z |
Cowingene Sample Release Reagent |
CE |
100 mL(10*10mL) |
| CW05021X |
Cowingene DNA/RNA Extraction kit |
CE |
48 tests/kit |
| CW08021Q |
Cowingene Sample Collection Kit |
CE |
1 test/kit |